A brief review of miniature flexible and soft tactile sensors for interventional catheter applications
Abstract
Interventional surgery has the advantages of small skin incisions, minimal blood loss, low postoperative infection and short recovery times, and thus has gradually become the preferred surgical approach over traditional open surgeries. Even though significant achievements have been made towards clinical applications, limitations still exist, among which the loss of the natural tactile perception of surgeons due to their indirect touch along the long catheter to the intervening human tissue is the crucial one. In recent years, researchers have dedicated great efforts to the development of advanced medical catheters with smart tactile perception ability, with considerable progress having been made. In this regard, we review the most recent developments of state-of-the-art miniature flexible and soft tactile sensors that are able to be integrated in the tip or on the side wall of medical catheters. We particularly focus on the sensing mechanisms, design requirements, device configuration and sensing performance of different types of sensors, as well as their application demonstration in synthetic anatomical models and in vivo animal experiments. After reviewing the representative research work, the challenges that still exist are summarized and the prospects toward future development are proposed.
Keywords
INTRODUCTION
Interventional surgery is a modern minimally invasive treatment. With the assistance of digital subtraction angiography and X-ray fluoroscopy, the surgeon introduces a specific precise medical instrument, such as a catheter, into the patient’s body by carrying out the operations of push, pull, twist and other manipulations in order to diagnose and treat the lesions on site[1]. At present, interventional surgery is mainly divided into four types, i.e., peripheral vascular intervention, neural intervention, tumor intervention[2] and cardiovascular intervention, all of which have achieved good clinical treatment results, effectively improving the quality of life and prolonging the survival time of patients[3]. In contrast, in traditional open surgery, the surgeon needs to anesthetize the patient, open a large wound in the appropriate part of the body, carry out the operation of probing, finding and cutting off the diseased part with a conventional large-sized surgical instrument and finally suture the wound. Traditional open surgery is prone to severe complications, including massive bleeding, postoperative infection and wound nonunion. Compared with traditional open surgery, interventional surgery with minimal invasiveness offers the comprehensive advantages of the reduction of intraoperative blood loss and tissue trauma, low risk of post-operative infection, small pain experienced by patients and short recovery time, and thus has gradually become the preferred approach in line with the trend of medical developments in the future[4].
Nevertheless, challenges still exist for surgeons when performing the operation of manipulating a thin and long medical catheter with flexibility and elasticity through the minimally invasive hole with indirect vision information from the digital subtraction angiography[5]. The major challenge lies in the difficulty in accurately positioning the catheter by the indirect sense of the contact between the far-away catheter tip and the intervening human tissue, especially when the tissue is continuously perturbed by movement during the operation[6]. Thus, a certain level of specific operating skills, gained from years of experience, is essential for experienced surgeons, which severely limits the popularization of interventional surgery in benefiting more patients. In order to improve the success rate of such operations, the surgeon is required to feel the hardness of the touching tissue through the long catheter and effectively evaluate the state of the patient's nerves or blood vessels. It would be a significant improvement to utilize a miniature tactile sensor that is able to be integrated in the tip or on the side wall of catheter to assist the surgeon to restore the sensitive tactile capability for a complete recognition and judgment of the touching condition surrounding the catheter. In addition, some lesions buried deeply underneath the surface of tissue are not easily found with advanced visual equipment but can be detected by sensitive touch[5].
Another challenge is the concern related to the safety of the surgeon who performs the operation closely beside the patient. In order to reduce the harm from X-ray irradiation, the surgeon has to wear heavy lead clothes for a long period during the whole operation process. The radiation dose is typically 1.41 mSv/a for peripheral vascular and cerebrovascular interventional surgery and 1.33 mSv/a for heart disease interventional surgery[7]. In addition, surgeons have to work under high attention with such heavy loads for long periods. Therefore, surgeons bear the occupational risks of both ionizing radiation and physical fatigue. It would be of great significance to develop an automatic catheter propulsion device or even interventional surgery robot to enable surgeons for a convenient and remote operation of the catheter. The advanced device or robot is capable of accurately locating the catheter to the target tissue site following a complex trajectory, performing continuous and stable catheter manipulation without vibrations and completing the whole interventional operation under the control of the surgeon. In such a case, the ionizing radiation and physical fatigue faced by surgeons can be remarkably reduced via the robotic-assisted interventional surgery process[8]. The key also lies in the development of miniature tactile sensors that can be integrated with medical catheters to acquire real-time and accurate tactile information between the catheter and its touching tissue as feedback for intelligent automatic control[9].
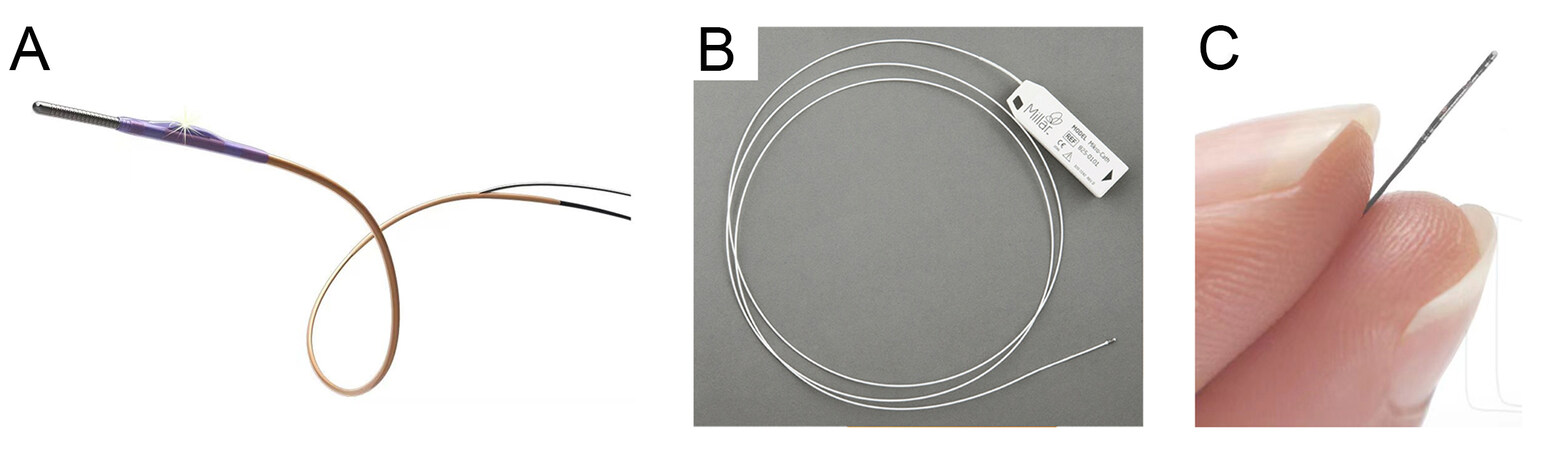
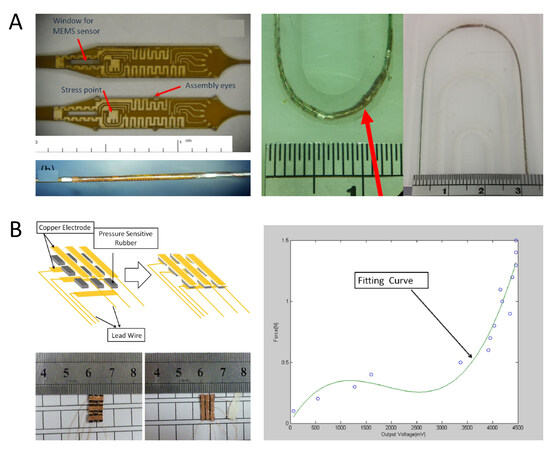
In industry, pioneering medical device companies have launched several advanced medical catheters equipped with miniature tactile sensors[10]. In China, InsightLifetech Co., Ltd., developed a blood flow measurement system known as fractional flow reserve (FFR) for clinical applications, which consists of a TRUEPHYSIO® pressure measuring catheter [Figure 1A][11-15] and VivoCardio® blood flow reserve fraction measuring equipment. The catheter is equipped with a miniature microelectromechanical system (MEMS) sensor (diameter of ~1.57 Fr) to offer the advantages of high accuracy (~93.4%), stable consistency (~99.6%) and low data drifting [reduced by 43% (> 0.03)]. By using the FFR measurement system, surgeons can judge the vascular health status and identify the stenosis lesions that cause the myocardial ischemia, so as to choose the best treatment plan for patients with the useful decision-making information. Millar Instruments Inc., in the United States, developed a pressure measuring catheter known as a Mikro-Cath for clinical applications, which is equipped with a miniature high-precision solid-state pressure sensor (diameter of ~3.5 Fr, Figure 1B). The pressure sensing catheter with a sensitivity of 5 µV/V/mmHg delivers an accurate measurement of the cardiovascular, respiratory, intra-compartmental and airway pressures in the human body in a short-term body contact (< 24 h). Scisense Inc., in Canada, presented a pressure measuring catheter equipped with a miniature tactile sensor on the side wall of the catheter tip (diameter of ~1.2-7.0 Fr, Figure 1C) for a variety of applications, which can measure the arterial and intracranial pressures of animals, such as mice, rats, rabbits, dogs and pigs, in in vivo animal experiments. The sensor with a sensitivity of 10 µV/V/mmHg provides a measurement range of -50 to + 300 mmHg within a temperature range of 32 to 42 °C.
Figure 1. Medical catheter products equipped with miniature tactile sensors. (A) TRUEPHYSIO® pressure measuring catheter from InsightLifetech Co., Ltd. (Available from: http://beixin.paiky.com.cn/product/122.html). (B) Mikro-Cath pressure measuring catheter from Millar Instruments Inc. (Available from: https://millar.com/Clinical/MikroCath/#Cardiovascular-Pressures). (C) Pressure measuring catheter from Scisense Inc. (Available from: http://www.bandetek.com/templates/consulting_008_1/second_14_148.html).
Currently, commercial pressure measuring catheter products mostly adopt the piezoresistive sensing mechanism, due to its simple sensing structure with good reliability. However, tactile sensors based on piezoresistive materials generally have the main drawback of performance drifting when the temperature changes significantly. In addition, the miniature tactile sensors for medical catheter products are mainly silicon-based MEMS device and thus are fabricated by using the standard photolithography technique that requires high-precision equipment and high-standard cleanroom facilities, leading to a high cost that is not promising towards mass production. Furthermore, the bulky and rigid formation of the sensor device may not be fully suitable for non-invasive or less invasive interventional surgery. Thus, miniature tactile sensors with high sensing performance, flexible mechanical properties and simple fabrication process still require further research and development.
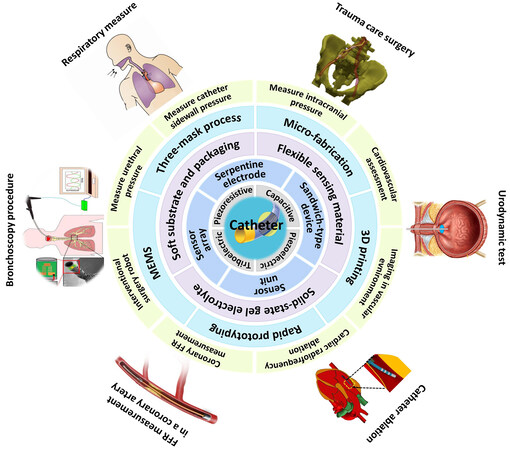
In academia, researchers have dedicated significant effort into researching new types of miniature tactile sensors with regards to the sensing mechanism, device structure, functional materials, fabrication method and potential application direction [Figure 2], with considerable progress having been made in recent years. This review aims to systematically summarize such research progress with the following structured contents. First, different working principles and device structures of tactile sensors are briefly introduced with focus on the comparison of their advantages and disadvantages towards different application needs. Second, the specific sensing requirements imposed by medical catheters towards the specific intervening surgery scenario are outlined. Third, recent representative research work is introduced in detail by classification of their sensing mechanism regarding device design, material usage, device fabrication and application demonstration. Finally, the conclusions and existing challenges are presented and future developments are proposed.
Figure 2. Schematic showing the research content of miniature tactile sensors for interventional catheters, including the sensing mechanism, device structure, functional material, fabrication method and potential application direction. Figures of “Bronchoscopy procedure” quoted from Gil et al.[11], “Respiratory measure” quoted from Kawaoka et al.[12], “Trauma care surgery” quoted from
WORKING PRINCIPLE AND DEVICE CONFIGURATION OF TACTILE SENSORS
A tactile sensor, also known as a pressure sensor, is a device that converts external stimuli, such as force or pressure, into measurable electrical signals caused by deformation of the sensing structure in the
Piezoresistive tactile sensors are mainly composed of a conductive layer fabricated on a substrate, in which the resistance of the conductive layer is changed upon deformation. The resistance of the piezoresistive material is determined by the formula of R = ρL/A, where R is the overall resistance, ρ is the resistivity, L is the length along the electron transportation direction and A is the cross-sectional area. When exerted by an external force, the piezoresistive layer is deformed, resulting in a change in the overall resistance. For example, elongation of the piezoresistive layer leads to increased L and decreased A, resulting in increased R. Piezoresistive sensors have the advantages of a simple and reliable device structure, easy device fabrication and convenient resistance signal acquisition. However, disadvantages exist due to the intrinsic electron transportation behavior, including relatively low sensitivity, large hysteresis of resistance change response to pressure and performance drifting upon temperature change. Compared with conventional bulky and rigid piezoresistive sensors, researchers have recently developed flexible piezoresistive sensors by using one-dimensional materials, such as silver nanowires[31], silicon nanowires[32] and carbon nanotubes[33], and two-dimensional materials, such as graphene[34], to form a network-type conductive layer on a flexible polymer substrate. The conductive connection within the network is still well retained when the substrate is bent or stretched, illustrating its potential for wearable or implantable sensing applications[35-37].
Capacitive tactile sensors usually adopt a sandwich-type device structure, i.e., a pair of parallel conductive electrodes sandwiching a deformable dielectric layer in the middle. The capacitance of the capacitive device is determined by the formula of C = εA/d, where C is the overall capacitance, ε is the dielectric constant, A is the electrode overlapping area and d is the distance between the two electrodes. When exerted by an external force, the dielectric layer is deformed, resulting in the change of the overall capacitance. For example, when compressed, the separation distance between the two electrodes is decreased and thus the capacitance is increased[38]. Capacitive tactile sensors have the advantages of high sensitivity, low power consumption and simple detection circuit design. In addition, they excel at measuring static force[39]. However, the absolute capacitance value of conventional capacitive tactile sensors is small, which is easily surpassed by the parasitic capacitance of human or electromagnetic waves in the environment. Therefore, for wearable or implantable applications, where heavy interference with humans exists, complicated signal acquisition and post-treatment circuit is highly required and it is still difficult to accurately measure subtle pressures. Recently, Pan and co-workers proposed the supercapacitive sensing mechanism by utilizing an iontronic material as the middle layer to form electric double-layer capacitors (EDLCs) at the electrode-electrolyte interface. The specific capacitance per areal unit of the iontronic supercapacitive sensor is observed to be 1000 times greater than that of a conventional electrostatic capacitive sensor, leading to the advantages of high sensitivity, broad detection range and high noise immunity[40]. By utilizing conductive nanomaterials and flexible polymer materials, researchers have also developed various kinds of flexible capacitive and supercapacitive tactile sensors with enhanced sensing performance through electrode, dielectric or electrolyte structuring towards wearable or implantable sensing applications[39].
Piezoelectric tactile sensors utilize a specific piezoelectric material [e.g., lead zirconate titanate and polyvinylidene fluoride (PVDF)] as the functional material, which generates a voltage when deformation of the crystal structure occurs as a result of an external force. Due to the intrinsic dynamic response behavior, piezoelectric tactile sensors are widely used in dynamic pressure detection, such as sliding and vibration. Considering the leakage problem, they are not capable of accurately measuring the static force. In addition, the sensing performance of piezoelectric sensors is easily disturbed by temperature, which limits their application in harsh environments of high temperatures and pressures. Triboelectric tactile sensors convert mechanical energy into electricity by using the coupling effect between triboelectrification and electrostatic induction through the contact separation or relative sliding between two functional materials that have the opposite tribopolarity[41]. Similar to piezoelectric tactile sensors, triboelectric tactile sensors excel at dynamic pressure detection. In addition, by taking advantage of electricity generation through continuous movements, advanced self-powered tactile sensors are developed based on triboelectric materials. However, due to the inert response to a static mechanical load, triboelectric tactile sensors fail in measuring static pressure[42]. In addition, due to the elegant nanowire structure of the electrodes and frequent friction movement, triboelectric sensors can be easily worn with a decay in the sensing performance, which is therefore not suitable for long-term use.
Table 1 summarizes the working principles of the four widely studied tactile sensors with superior performance and usefulness for many applications[43-51]. It should be noted that each type of tactile sensor upon its sensing mechanism has its own advantages and disadvantages, so a good selection of sensor type and fabrication of the sensor device should be considered for specific practical applications by different requirements. The piezoresistive sensors are easy to operate with simple signal conditioning circuit but suffer from performance drift when the temperature changes excessively. In addition, the thermal noise generated by the resistance measurement principle may also have a substantial impact on the final signal quality in a dynamic and noisy environment[52]. Capacitive tactile sensors have high sensitivity and a wide measurement range for static pressure detection and can also be made into an array for spatial pressure distribution measurements. However, they are easily influenced by the environmental parasitic capacitance and electromagnetic noise, so supercapacitive tactile sensors with increased capacitance should be further developed. Both of the piezoelectric and triboelectric tactile sensors excel at the dynamic pressure detection, making them suitable for measuring frequent movements. However, they do not respond well to a static mechanical load, therefore limiting their application in any absolute pressure measurement.
Basic principles, characteristics and applications of tactile sensors
| Classification | Piezoresistive | Capacitive | Piezoelectric | Triboelectric |
| Schematic diagram of working mechanism | 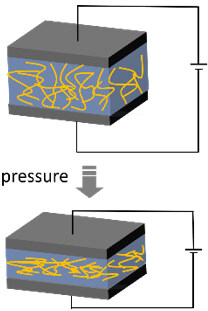 | 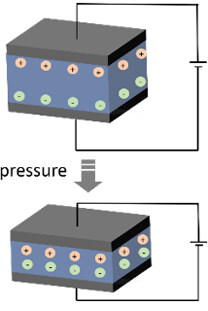 | 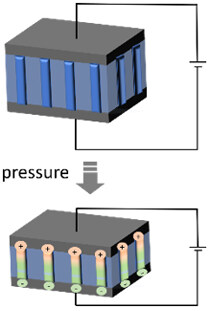 | 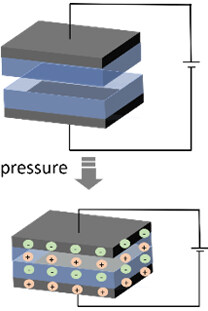 |
| Sensing principles | External force causes deformation of the conductor, leading to variation of the contact within the internal conducting network. The changed resistance or current can be measured | External force causes deformation of the capacitor, leading to variation of either the distance or overlapping area between the two electrodes. The changed capacitance can be measured | External force causes deformation of the piezoelectric material, leading to the occurrence of electrical dipole moments. The generated net electrical charges on the surface can be measured | When two kinds of materials with different triboelectric polarities are rubbed with each other, opposite electrical charges are induced on both sides of surfaces, which can be measured |
| Merits | Simple device structure, easy device fabrication and convenient signal acquisition | High sensitivity, low power consumption and suitable for static sensing | Wide working range and high sensitivity for dynamic sensing | High sensitivity for dynamic sensing and can be self-powered |
| Demerits | Influenced by temperature, as well as comparatively high-power dissipation | Disturbed by parasitic capacitance and electromagnetic noise, as well as a hysteresis effect | Susceptibility to temperature and not suitable for static sensing | Not suitable for static sensing and poor cycle stability |
| Exampled applications | Interactive electronics[44,45] and artificial peripheral neural systems[46,47] | Haptic sensation of soft robots[48] and electrodermal devices[49] | Vital physiological pressure measurements[50] | Tactile electronic skin with self-powered capability[51] |
SPECIFIC APPLICATION REQUIREMENTS FOR TACTILE SENSORS TO BE INTEGRATED WITH INTERVENTIONAL CATHETERS
With the development of medical technology in recent years, interventional surgery has become an effective method to treat cardiovascular and cerebrovascular diseases, tumors and other internal diseases. After puncture and intubation, the patient is examined by pathology and cytology, and then treated by the surgeon with a medical catheter for drug perfusion and vascular embolization under the assistant of the medical imaging technology. A catheter is a specific precise medical instrument that is designed and used to enter the vascular cavity through arterial puncture for diagnosis and treatment. The catheter also plays a vital role in many other medical procedures, such as angiography, ablation, minimally invasive surgery[53] and urology[54]. The timely and accurate feedback of the contact force with physical characteristics between the catheter tip and the tissue of the intervening organ is crucial for the surgeon’s operation, and it would be a significant improvement if miniature tactile sensors can be integrated on the catheter to improve the surgical accuracy, success rate and efficiency[55]. In addition, the recent fast development of advanced functional nanomaterials and elastic polymer materials has made it possible to fabricate flexible and soft sensor devices, which possess the merits of being less invasive to human tissue. By utilizing the micro-fabrication-related technique, the fabrication of miniature, flexible and soft tactile sensors that are ready to be integrated on medical catheters become feasible.
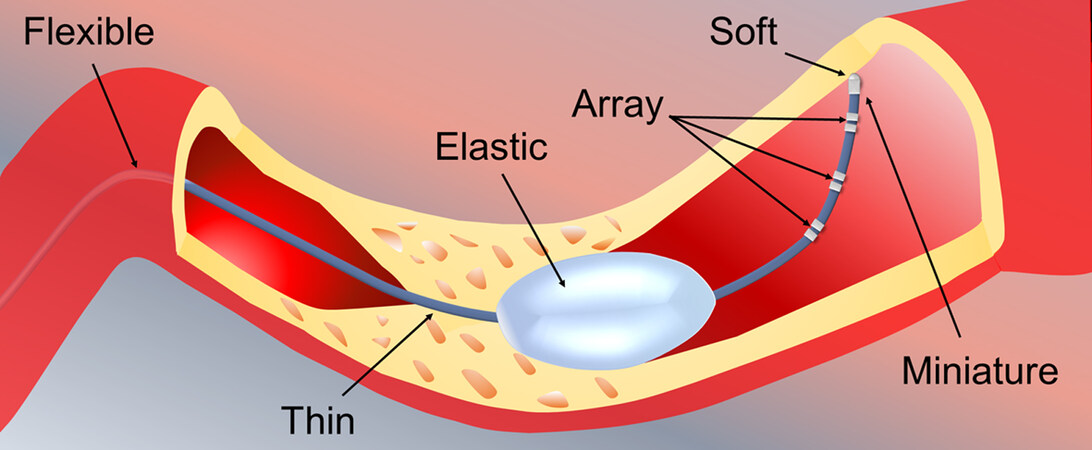
To develop such tactile sensors for interventional catheters, in addition to the selection of the sensing mechanism and the design of the device configuration, comprehensive factors, such as form factor, mechanical properties and sensing behavior, must be considered in order to meet the specific requirements imposed by the intervening application scenario [Figure 3].
First, the tactile sensor must be miniature, thin and able to be tightly integrated in the tip or on the side wall of the catheter in order not to increase the thickness and size of the original catheter. In the field, the Charriere’s French (Fr) gauging system is widely used to define the catheter size, where one Fr denotes a 0.3 mm outer diameter of the catheter tube. The size of a catheter ranges from 24 down to 1 Fr (~7.2 to 0.3 mm), depending on different applications[56]. Thus, tactile sensors should be fabricated at the millimeter or even sub-millimeter scale.
Second, the material used to construct the tactile sensor is highly required to be flexible and elastic in order to effectively avoid unnecessary invasion to the internal human tissue. The flexibility enables the tactile sensor to accommodate the curving movement of the catheter along the intervening vascular cavity. The elasticity makes the tactile sensor to be soft that is non-invasive to human tissue and can even be integrated on the balloon of the ablation catheter.
Third, the functional material used to build the tactile sensor should be inert to X-ray radiation during the angiography process[56] and the packaging material must be biocompatible with the biological tissue and non-toxic to the human body. Regarding the integration layout, a single tactile sensor can be mounted on the tip of the catheter to acquire the one-point pressure information between the front end of the catheter and the touching tissue, while an array of multiple tactile sensors can also be assembled at intervals on the side wall along the perimeter or length of the catheter tube in order to detect the spatial multi-point pressure information surrounding the catheter. Regarding the sensing performance, the tactile sensor should be able to offer a detection limit of ~0.01 N (1 g) and measurement range of ~0.0-1.5 N (0-150 g) with more than 95% linearity and less than 10% hysteresis[28,57].
RESEAECH PROGRESS OF MINIATURE TACTILE SENSORS FOR INTERVENTIONAL CATHETERS
Pressure sensing is of significant importance in diagnosing and treating critical biomedical implant conditions. In this regard, a variety of tactile sensors with a tiny form factor for interventional catheters have been extensively explored by researchers based on different sensing mechanisms, including piezoresistive, capacitive, supercapacitive and piezoelectric sensors. Significant efforts have been dedicated in achieving miniature tactile sensors with a simple structure, high sensitivity, a broad measurement range, stable reliability and feasible integration with commercial medical catheters.
Piezoresistive tactile sensors
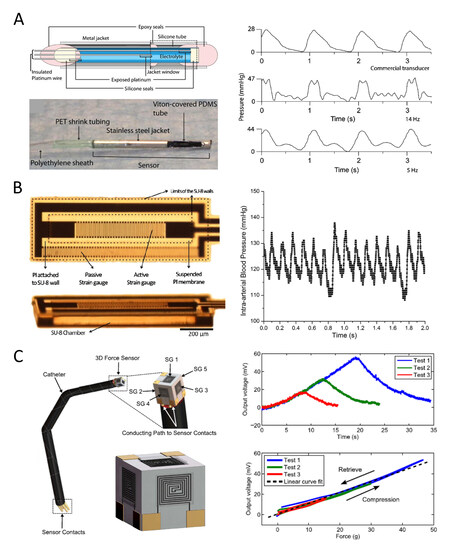
In order to find an alternative approach to measure the arterial blood pressure, Tan et al. developed an implantable electrolyte conductance-based pressure sensing catheter based on an elastic deformable tube filled with electrolyte solution, as shown in Figure 4A[58]. This catheter works by measuring the conductivity change of the electrolyte caused by the pressure-induced tube deformation. The sensor has a diameter of
Figure 4. Miniature piezoresistive tactile sensors for catheters. (A) A tube-type tactile sensor consisting of an aqueous 50 mM KCl electrolyte as a conductor (left part) and its response sensing curve filtrated at 14 Hz (middle, right plot) and 5 Hz (bottom, right plot) compared to a commercial transducer (top, right plot) in an in vivo test[58]. (B) A PI/SU-8 MEMS gauge pressure sensor (left part) and its response sensing signal curve of carotid arterial blood pressure and heart rate in a male mouse during inhalation of isoflurance (right plot)[59]. (C) A PEDOT:PSS/PDMS MEMS force sensor for triaxial catheter contact force measurements (left part) and its output voltage with respect to time (top, right plot) and sensitivity (bottom, right plot)[61]. MEMS: Microelectromechanical systems; PDMS: polydimethylsiloxane.
Based on the flexible, electrically insulating, thermal stable and biocompatible polyimide (PI) through the micromachining fabrication process with the use of negative epoxy-based photoresist SU-8,
By changing the pressure-based sensing mechanism to a force-based sensing mechanism that improves the signal resolution at a higher contact angle between the catheter and blood vessel, Huang et al. developed a miniature tactile sensor for a cardiovascular catheter[60]. The sensor consists of a compressible diaphragm with a highly sensitive strain gauge (thickness ~0.2 mm, height ~4 mm and diameter ~7 mm), a contact part (304 SS), a shelter part (304 SS) and an elastic rubber ring. When the suction head compresses the diaphragm, different resistance values are generated due to structural changes. The strain gauge then collects the changing electrical signals to reflect the applied pressure. The diaphragm with a 0.2 mm thickness was found to obtain a sensing force larger than 100 mN. Tested under three different experimental fiber placement conditions, the fiber photoacoustic imaging was successfully produced, although the signal was weak, thereby verifying the feasibility of the photoacoustic imaging in a human vascular environment.
In addition to integrate one single miniature piezoresistive tactile sensor on the catheter tip,
Electrostatic capacitive tactile sensors
For the FFR measurement of coronary arteries, Van den Driesche et al. integrated a MEMS capacitive pressure sensor on the outer wall of the catheter, as shown in Figure 5A[15]. The sensor was designed with a peculiar long and narrow oblong shape, which doubles the sensitivity over traditional circular membrane sensors with equal area and deflection and thus improves the robustness of the sensor under external forces. The pressure sensor is specified for -30 to 300 mmHg relative blood pressure (absolute 570 to 1125 mm Hg with a barometric range of 600 to 825 mmHg). The miniature MEMS sensor and application-specific integrated circuit chip dies were successfully integrated in a 2 Fr catheter through the flip-chip interconnection micro-assembly technique. Track, kink and tensile tests were carried out to evaluate the pressure measuring catheter performance.
Figure 5. Miniature capacitive tactile sensors for catheters. (A) A MEMS capacitive pressure sensor (left part) and the kink test shows that the assembly could pass 15 and 12.5 mm radii but failed at a 7.5 mm radius (right plot)[15]. (B) A capacitive sensor array on catheter sidewall (left part) and fitting curve of the sensing data (right plot)[62]. MEMS: Microelectromechanical systems.
In addition, to integrate one single miniature capacitive tactile sensor with the catheter, Guo et al. developed a 3 × 3 tactile sensor array with a pressure sensitive rubber, which could be installed on the catheter side wall to detect the contacting force with the blood vessel, as shown in Figure 5B[62]. The single pressure sensitive rubber had dimensions of
Supercapacitive tactile sensors
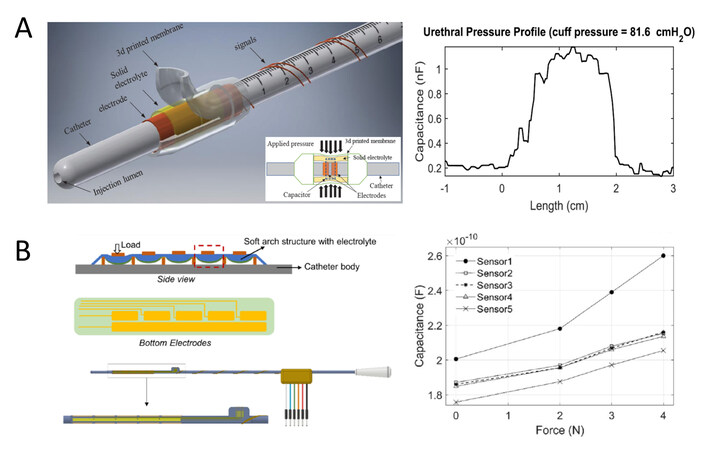
In order to overcome the parasitic capacitance noise from the environment, Ahmadi et al. developed a supercapacitive pressure transducer for the urethral catheter, as shown in Figure 6A[63]. The sensor was fabricated by putting a solid ionic electrolyte film on top of two gold coated copper electrodes. The solid electrolyte was bonded to a 3D printed soft balloon to ensure sufficient stiffness to undergo the repeated compressing and releasing process. The two electrodes were designed to have a 200 μm gap between each other, so when the outside soft balloon was compressed by an external force, the solid electrolyte would have a firm contact with the two electrodes and a supercapacitor device was formed along the connection of the electrode/electrolyte/electrode structure. The capacitance of the supercapacitor changes according to the effective contact area between the electrolyte and electrodes. The whole sensor was built on the side wall of a 6 Fr urethral catheter and was evaluated in the in-vitro experiment to observe a sensitivity of 10 pF/cmH2O.
Figure 6. Miniature supercapacitive tactile sensor for catheters. (A) A supercapacitive pressure sensor based on a solid electrolyte (left part) and its response sensing signal curve in the in-vitro test with a cuff pressure of 81.6 cmH2O (right plot)[63]. (B) An array of distributed iontronic force sensor (left part) and its capacitance responses to the force inside the extracted sheep urethra (right plot)[14].
To measure the force distribution in the urethra during clinical diagnosis in patients, Zhang et al. developed an instrumented urethral catheter with a distributed array of five iontronic force sensors along the length of the catheter, as shown in Figure 6B[14]. The soft substrate with electrodes was assembled on a flat portion of the 3D printed 7 Fr catheter body and a chamber was also printed with a combination of both hard and soft materials to host the electrolyte and perform deformation under loads. The electrolyte was made solid state by brushing an ionic liquid and photocurable polymer on filter paper to achieve both excellent ionic and mechanical properties. The vitro load cell experiment, cuff test, in-water test and extraction bladder test were carried out to evaluate the performance of the pressure sensing catheter. The sensor was observed to have a high sensitivity of 30-50 nF/N, which is more than 1000 times that of traditional capacitive sensors on similar-sized catheters. Furthermore, it could effectively resist the influence of parasitic capacitance.
Piezoelectric tactile sensors
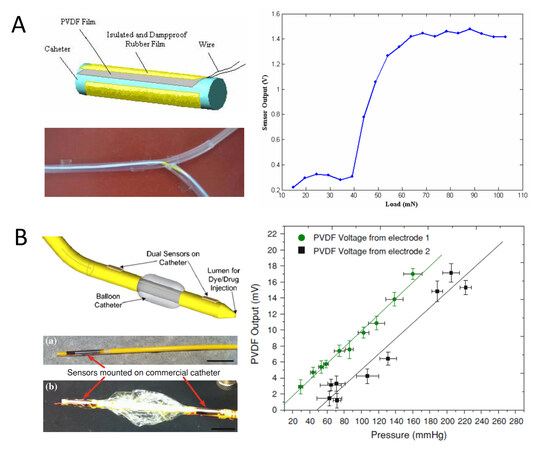
Based on the typical piezoelectric polymer of PVDF, Feng et al. developed a miniature tactile sensor to measure the pressure on the side wall of a catheter, as shown in Figure 7A[64]. The sensing system is composed of the catheter, PVDF film, insulated and damp proof rubber film and output wire, and has a diameter of 2.5 mm and a length of 20 mm. The sensor works based on the piezoelectric effect of the PVDF, i.e., an equal number of opposite polarity electric charges on both sides of the material that is accumulated when a load is applied. Electrical tests showed that the sensor had a good dynamic response and a linear output in a certain load range. A simulative experiment in the artificial blood vessel was also performed to verify the application performance of the pressure measuring catheter.
Figure 7. Miniature piezoelectric tactile sensors for catheters. (A) A tactile sensor based on a PVDF film (left part) and its output voltage with respect to applied load (right plot)[64]. (B) A pressure sensor based on PVDF-TrFE copolymer film (left part) and its output voltage with respect to applied pressure (right plot)[13]. PVDF: Polyvinylidene fluoride.
Based on the piezoelectric copolymer of poly(vinylidene fluoride-tetrafluoroethylene) P(VDF-TrFE), Sharma et al. developed a flexible thin-film pressure sensor for smart catheter applications, as shown in Figure 7B[13]. Two types of sensors with single- and dual-membrane geometries with bottom electrode sizes of 2 mm × 10 mm and 5 mm × 10 mm were fabricated to optimize the signal output while maintaining a compact form factor for catheter integration. The sensor was constructed on a Kapton tape (thin polyimide film) as a substrate with patterning of the sensor structure of the bottom Al electrode, middle polymer layer and top Cu electrode by using the standard photolithography process. The sensor was measured to have a broad pressure range of 0-300 mmHg with an average sensitivity of 99 μV/mmHg (four times higher than a commercial pressure sensor) and fast response time of 0.26 s (fivefold shorter than a commercial pressure sensor). Prototype sensors mounted on a commercial medical catheter and either side of a balloon were also demonstrated.
Application demonstration in synthetic anatomical models and in vivo animal experiments
After various types of miniature tactile sensors based on different sensing mechanisms have been successfully fabricated with small and compact form factors for good integration with thin catheters, mechanical and electrical measurements are firstly performed to test the sensing performance upon mechanical stimuli. The developed pressure measuring catheters need to be evaluated in the synthetic anatomical models and even in vivo animal experiments to further verify their practical functions towards final clinical applications.
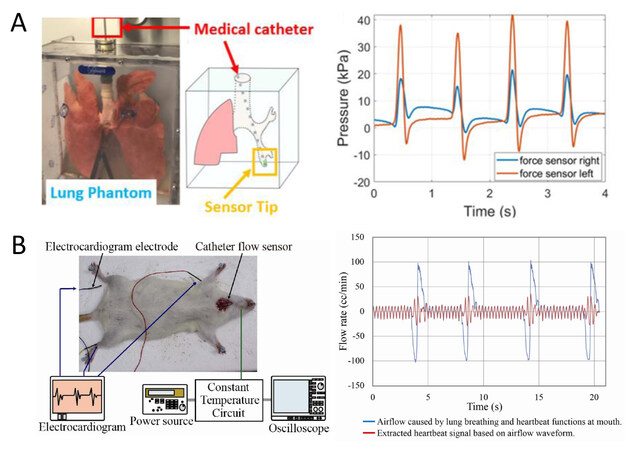
Gil et al. fabricated a miniature force sensor made of P(VDF-TrFE) and integrated it into the tip of a medical catheter for intraluminal pressure monitoring[11]. The sensor shows a pressure range of below
Figure 8. Application demonstration of catheters equipped with miniature tactile sensors. (A) Lung model used to test a medical catheter with bilateral pressure sensing capability (left part) and corresponding pressure signals obtained by the two sensors inside the left lobar branch of the lung (right plot)[11]. (B) In vivo measurement of the oral airflow in rats using a flow sensing catheter (left part) and extracted heartbeat signals from the airflow waveform at a rat’s mouth (right plot)[12].
Kawaoka et al. developed a catheter flow sensor with an outer diameter of 1.8 mm by using the MEMS fabrication technique[12]. The MEMS thermal flow sensor was firstly fabricated by patterning two Au/Cr metal heaters on a flexible PI substrate (total sensor size ~2.8 mm × 5.5 mm) and then inserted into two heat-shrinkable tubes, followed by integration in a catheter to detect the air flow at the mouth. The sensor shows a sufficiently small response time of 68.5 and 57.5 ms in rise and fall modes, respectively, which ensures a dynamic characteristic of the flow sensing. As shown in Figure 8B, the animal experiment was carried out by directly inserting the catheter flow sensor into the airway of a rat under heavy anesthesia. The airflow signal caused by the heartbeat was clearly observed in the time interval between the signals caused by lung breathings, from which the frequency information was analyzed with coincidence to that of the electrocardiogram graph.
Table 2 summarizes the working principles, sensing materials, device structures, miniature dimensions, sensing performance and application directions in the recently reported research work on miniature flexible and soft tactile sensors for interventional catheters. Various kinds of sensing materials, such as metals, conducting polymers, liquid or gel electrolyte and PVDF have been used to construct the typical four types of tactile sensors with small dimensions on the flexible polymer substrate, including PDMS and PI. The miniature tactile sensors can be successfully integrated on medical catheters for demonstrated proof-of-concept experiments towards various interventional applications.
Principles, materials, structures, dimensions, performance and applications of miniature tactile sensors for interventional catheters reported in the literature
| Working principle | Sensing material | Device structure | Miniature dimension | Sensing performance | Application direction | Ref. |
| Piezoresistive | Conductive KCl electrolyte | Elastic tube filled with electrolyte | Diameter: 2.3 mm Length: 30 mm | Sensitivity: 1.42 mV/mmHg Detection range: 250 mmHg | Measure intracranial pressure | [58] |
| Micro-machined metal wires | MEMS electrode on PI membrane | 0.4 mm ×1.2 mm | Sensitivity: 2.78 μV/mmHg | Cardiovascular assessment | [59] | |
| Stainless steel diaphragm | Cylindrical shape | Diameter: 7 mm Thickness: 0.2 mm Height: 4 mm | Sensitivity: ~2.5 × 10-8 V/mN Detection range: 100 to | Imaging in vascular environment | [60] | |
| PEDOT:PSS strain gauge | MEMS electrode on PDMS substrate | 2000 μm × 2000 μm × 1100 μm | Sensitivity: ~1 mV/g Detection range: 0 to 40 g | Cardiac radiofrequency ablation | [61] | |
| Electrostatic capacitive | Silicone gel on diaphragm | MEMS capacitor Sensor unit | Length: 2.45 mm 2 Fr catheter | Detection range: 570 to 1125 mmHg | Coronary FFR measurement | [15] |
| Sensitive rubber with copper electrode films | Capacitor 3 × 3 array | 3 mm × 1.5 mm × 0.5 mm | Output of 172 mV equals to 0.12 N | Interventional vascular surgery robot | [62] | |
| Supercapacitive | Solid ionic electrolyte on top of two gold coated copper electrodes | Planar capacitor Sensor unit | 6 Fr catheter | Sensitivity: 10 pF/cmH2O | Measure urethral pressure | [63] |
| Solid ionic gel with copper tape electrodes | Capacitor 5 units in a row | 7 Fr catheter | Sensitivity: 30 to 50 nF/N | Simultaneously measure pressure at multiple locations in the urethra | [14] | |
| Piezoelectric | PVDF film | Film wrapping on catheter | Diameter: 2.5 mm Length: 20 mm | Linear behavior between 40 to 60 mN | Side wall pressure of cardiovascular surgical catheter | [64] |
| PVDF-TrFE co-polymer film | Thin-film sensors on Kapton film | 2 mm × 10 mm 5 mm × 10 mm | Sensitivity: 99 μV/mmHg Detection range: 0 to Response time: 0.26 s | Measure catheter sidewall pressure in vascular test | [13] |
CONCLUSION AND OUTLOOK
Compared with traditional open surgery, interventional surgery is a modern minimally invasive treatment, offering comprehensive advantages of small skin incisions, minimal blood loss, low postoperative infection and short recovery times. Currently, there are still two key issues: (1) the loss of direct contact sense of the catheter tip with the touching human tissue makes it difficult for the surgeon to accurately manipulate the thin and long catheter, thus years of training is highly required by surgeons, which limits the popularization of the interventional surgery to benefit more patients; (2) due to the using of digital subtraction angiography and X-ray fluoroscopy to assist the vision of the actual catheter movement in the human body, surgeons bear the occupational risks of both ionizing radiation and physical fatigue through doing the elegant operation for a long time with high attention by wearing the heavy lead clothes. Equipping the catheter with miniature tactile sensors in the tip or on the side wall can directly acquire real-time and accurate pressure information surrounding the catheter, which facilitate the surgeon to perform a more accurate manipulation and benefit in inventing the automatic catheter propulsion device or interventional surgery robot. Therefore, the development of miniature tactile sensors that can be integrated into the medical catheters has become greatly significant.
In recent years, significant progress has been made in both industrial and academic fields. In industry, pioneering medical device companies have launched several types of pressure sensing catheter products, but their performance needs to be further improved and the manufacturing costs need to be largely reduced. In academia, a considerable amount of research has been done to explore new types of miniature tactile sensors that are able to meet the multiple requirements of compact device configurations, small form factors, mechanical flexibility and elasticity and excellent sensing performance, which are imposed by the specific application scenario of moving the thin catheter in the long, narrow and curved cavity in the human body. Four types of miniature tactile sensors based on the piezoelectric, capacitive, supercapacitive and piezoelectric sensing mechanism have been researched by well selection of advanced functional materials and implementation of microfabrication-based techniques. They can be successfully mounted either in the catheter tip or on the side wall and integrated either as a single sensor unit to monitor one-point pressure of the catheter tip or as a sensor array to detect multi-point pressure distribution surrounding the catheter. The developed pressure sensing catheters are firstly evaluated by mechanical and electrical measurements to observe a workable sensing performance that meets the application needs, and then tested in the synthetic anatomical models and even in vivo animal experiments to verify their feasibility towards future clinical applications.
Even though great progress has been achieved, challenges still exist and further research is needed in the following aspects:
(1) Sensing mechanism and device configuration: piezoresistive, capacitive and piezoelectric miniature tactile sensors for medical catheters are widely researched with their own advantages and disadvantages. Piezoresistive sensors are simple in structure and easy to fabricate, but their sensing characteristics are highly affected by temperature and specific circuits for temperature changing compensation are needed for implant applications. Capacitive sensors are good at measuring the static force and easy to construct sensor arrays for multi-point pressure measurement, but their capacitance is easily disturbed by the parasitic capacitance of the human body and electromagnetic waves in the environment. Piezoelectric sensors are good at detecting dynamic pressure with high sensitivity, but they fail to measure the static force. It is highly required to develop tactile sensors based on new sensing mechanisms to achieve a breakthrough in sensing performance. The invention of the iontronic supercapacitive sensing mechanism to obtain a remarkably increased capacitance compared to traditional electrostatic capacitive sensors to overcome the environmental interfering sets a good example.
(2) Functional sensing materials: to meet the flexibility and softness requirements, nanofilms of metal and conducting polymers are patterned on flexible and elastic polymer substrate to fabricate flexible miniature tactile sensors. In addition, for iontronic tactile sensors, solid-state electrolytes formed by introducing polymer chains as mechanical frameworks have been invented to avoid liquid leakage issue. In the future, more types of advanced functional materials, such as silver nanowires, CNTs, graphene, MXenes and hydrogels, which are already widely investigated in wearable electronics could be introduced to the development of miniature tactile sensors for medical catheters.
(3) Fabrication of the sensor device and its integration with the catheter: miniature tactile sensors are mostly fabricated by using the microfabrication technique and delicately assembled in the tip or on the side wall of the catheter with extremely careful attention. The process of firstly fabricating individual sensor and then integrating on catheter is complicated with relatively low yield rate, and thus the cost is too high for its use in a broad range. Thus, it is necessary to explore new fabrication and integration approach and it would be great if the miniature tactile sensors could be in-situ fabricated on a catheter.
(4) Sensing performance test and application verification experiment: electrical and mechanical tests are carried out to measure the key sensing performance parameters including sensitivity, measurement range and response time. While other properties, such as sensing hysteresis, cycling stability, durability and reliability in changing environment should also be comprehensively tested and discussed before practical application. In addition to relatively simple demonstrations of experiments evaluating basic functions of developed pressure measuring catheters in simulative models, more in vivo animal and even human experiments should be performed with dada collection to fully prepare for the final clinical application.
DECLARATIONS
AcknowledgmentsThis work was supported by the National Natural Science Foundation of China (No. 61871173) and partially by the Fund for Innovative Research Groups of Natural Science Foundation of Hebei Province (No. A2020202002).
Authors’ contributionsInitiated the reviewing idea and outlined the manuscript structure: Meng C
Did the literature review and wrote the manuscript draft: Li Y, Wang P
Participated in figure drawing: Chen W, Zhang L
Were involved in the discussion and revised the manuscript: Meng C, Guo S
All authors have read the manuscript and approved the final version.
Availability of data and materialsNot applicable.
Financial support and sponsorshipThis work was supported by National Natural Science Foundation of China (No. 61871173) and partially by the Fund for Innovative Research Groups of Natural Science Foundation of Hebei Province (No. A2020202002).
Conflicts of interestAll authors declared that there are no conflicts of interest.
Ethical approval and consent to participateNot applicable.
Consent for publicationNot applicable.
Copyright© The Author(s) 2021.
REFERENCES
1. Jing X, Ken C, Xiangdong Y, Ping L. Experimental study on accuracy of robot assisted interventional therapy system. Mech Des Manuf 2010;1:156-8.
2. Wang D, Dong R, Wang X, Jiang X. Flexible electronic catheter based on nanofibers for the
3. Yaling H. Chinese guidelines for percutaneous coronary intervention 2012 (simplified version). Chinese J Med Front (Electronic Edition) 2012;4:50-9. Available from: https://yxqy.cbpt.cnki.net/WKB3/WebPublication/paperDigest.aspx?paperID=289AEDE4-A21E-4FE7-8F17-2DD7DC9F8E95# [Last accessed on 1 June 2022]
4. Slotta JE, Kollmar O, Ellenrieder V, Ghadimi BM, Homayounfar K. Hepatocellular carcinoma: Surgeon’s view on latest findings and future perspectives. World J Hepatol 2015;7:1168-83.
5. Puangmali P, Althoefer K, Seneviratne LD, Murphy D, Dasgupta P. State-of-the-Art in force and tactile sensing for minimally invasive surgery. IEEE Sensors J 2008;8:371-81.
6. Sitaramgupta VVSN, Padmanabhan D, Rao PSM, Pandya HJ. Force sensing technologies for catheter ablation procedures. Mechatronics 2019;64:102295.
7. Qian Q. Analysis of radiation environmental impact of digital subtraction angiography. Dig Technol Appl 2020;38:3.
8. Schostek S, Schurr MO, Buess GF. Review on aspects of artificial tactile feedback in laparoscopic surgery. Med Eng Phys 2009;31:887-98.
9. Bandari N, Dargahi J, Packirisamy M. Tactile sensors for minimally invasive surgery: a review of the state-of-the-art, applications, and perspectives. IEEE Access 2020;8:7682-708.
10. Trevino RJ, Jones DL, Escobedo D, et al. Validation of a new micro-manometer pressure sensor for cardiovascular measurements in mice. Biomed Instrum Technol 2010;44:75-83.
11. Gil B, Li B, Gao A, Yang GZ. Miniaturized Piezo force sensor for a medical catheter and implantable device. ACS Appl Electron Mater 2020;2:2669-77.
13. Sharma T, Aroom K, Naik S, Gill B, Zhang JX. Flexible thin-film PVDF-TrFE based pressure sensor for smart catheter applications. Ann Biomed Eng 2013;41:744-51.
14. Zhang Y, Ahmadi M, Timm G, Sezen S, Rajamani R. An instrumented urethral catheter with a distributed array of iontronic force sensors. Ann Biomed Eng 2021;49:149-61.
16. Tiwana MI, Redmond SJ, Lovell NH. A review of tactile sensing technologies with applications in biomedical engineering. Sens Actuators A Phys 2012;179:17-31.
17. Lee B, Kim J, Kim H, Kim C, Lee S. Low-cost flexible pressure sensor based on dielectric elastomer film with micro-pores. Sens Actuators A Phys 2016;240:103-9.
18. Pan L, Chortos A, Yu G, et al. An ultra-sensitive resistive pressure sensor based on hollow-sphere microstructure induced elasticity in conducting polymer film. Nat Commun 2014;5:3002.
19. Li RZ, Hu A, Zhang T, Oakes KD. Direct writing on paper of foldable capacitive touch pads with silver nanowire inks. ACS Appl Mater Interfaces 2014;6:21721-9.
20. Woo SJ, Kong JH, Kim DG, Kim JM. A thin all-elastomeric capacitive pressure sensor array based on micro-contact printed elastic conductors. Mater Chem 2014;2:4415-22.
21. Pan C, Dong L, Zhu G, et al. High-resolution electroluminescent imaging of pressure distribution using a piezoelectric nanowire LED array. Nat Photon 2013;7:752-8.
22. Wu W, Wen X, Wang ZL. Taxel-addressable matrix of vertical-nanowire piezotronic transistors for active and adaptive tactile imaging. Science 2013;340:952-7.
23. Persano L, Dagdeviren C, Su Y, et al. High performance piezoelectric devices based on aligned arrays of nanofibers of poly(vinylidenefluoride-co-trifluoroethylene). Nat Commun 2013;4:1633.
24. Bauer S, Gerhard-multhaupt R, Sessler GM. Ferroelectrets: soft electroactive foams for transducers. Phys Today 2004;57:37-43.
25. Wang ZL. From contact electrification to triboelectric nanogenerators. Rep Prog Phys 2021;84:096502.
26. Pan S, Zhang Z. Fundamental theories and basic principles of triboelectric effect: A review. Friction 2019;7:2-17.
27. Noh Y, Liu H, Sareh S, et al. Image-based optical miniaturized three-axis force sensor for cardiac catheterization. IEEE Sensors J 2016;16:7924-32.
28. Polygerinos P, Seneviratne LD, Razavi R, Schaeffter T, Althoefer K. Triaxial catheter-tip force sensor for MRI-guided cardiac procedures. IEEE/ASME Trans Mechatron 2013;18:386-96.
29. Polygerinos P, Ataollahi A, Schaeffter T, Razavi R, Seneviratne LD, Althoefer K. MRI-compatible intensity-modulated force sensor for cardiac catheterization procedures. IEEE Trans Biomed Eng 2011;58:721-6.
30. Nakagawa H, Kautzner J, Natale A, et al. Locations of high contact force during left atrial mapping in atrial fibrillation patients: electrogram amplitude and impedance are poor predictors of electrode-tissue contact force for ablation of atrial fibrillation. Circ Arrhythm Electrophysiol 2013;6:746-53.
31. Jin ML, Park S, Lee Y, et al. An Ultrasensitive, visco-poroelastic artificial mechanotransducer skin inspired by piezo2 protein in mammalian merkel cells. Adv Mater 2017;29:1605973.
32. Phan H, Kozeki T, Dinh T, et al. Piezoresistive effect of p-type silicon nanowires fabricated by a top-down process using FIB implantation and wet etching. RSC Adv 2015;5:82121-6.
33. Dinh T, Nguyen T, Phan H, et al. Electrical resistance of carbon nanotube yarns under compressive transverse pressure. IEEE Electron Device Lett 2018;39:584-7.
34. Shi G, Lowe SE, Teo AJ, et al. A versatile PDMS submicrobead/graphene oxide nanocomposite ink for the direct ink writing of wearable micron-scale tactile sensors. Appl Mater Today 2019;16:482-92.
35. Mu C, Song Y, Huang W, et al. Flexible Normal-tangential force sensor with opposite resistance responding for highly sensitive artificial skin. Adv Funct Mater 2018;28:1707503.
36. Sun B, McCay RN, Goswami S, et al. Gas-permeable, multifunctional on-skin electronics based on laser-induced porous graphene and sugar-templated elastomer sponges. Adv Mater 2018;30:e1804327.
37. Gong S, Schwalb W, Wang Y, et al. A wearable and highly sensitive pressure sensor with ultrathin gold nanowires. Nat Commun 2014;5:3132.
38. Lee H, Chung J, Chang S, Yoon E. Real-time measurement of the three-axis contact force distribution using a flexible capacitive polymer tactile sensor. J Micromech Microeng 2011;21:035010.
39. Frutiger A, Muth JT, Vogt DM, et al. Capacitive soft strain sensors via multicore-shell fiber printing. Adv Mater 2015;27:2440-6.
40. Nie B, Xing S, Brandt JD, Pan T. Droplet-based interfacial capacitive sensing. Lab Chip 2012;12:1110-8.
41. Wang ZL. Triboelectric nanogenerators as new energy technology for self-powered systems and as active mechanical and chemical sensors. ACS Nano 2013;7:9533-57.
42. Chang Y, Wang L, Li R, et al. First decade of interfacial iontronic sensing: from droplet sensors to artificial skins. Adv Mater 2021;33:e2003464.
43. Yang T, Xie D, Li Z, Zhu H. Recent advances in wearable tactile sensors: materials, sensing mechanisms, and device performance. Mater Sci Eng R Rep 2017;115:1-37.
44. Liao X, Wang W, Zhong L, Lai X, Zheng Y. Synergistic sensing of stratified structures enhancing touch recognition for multifunctional interactive electronics. Nano Energy 2019;62:410-8.
45. Liao X, Wang W, Wang L, et al. A highly stretchable and deformation-insensitive bionic electronic exteroceptive neural sensor for human-machine interfaces. Nano Energy 2021;80:105548.
46. Liao X, Song W, Zhang X, et al. A bioinspired analogous nerve towards artificial intelligence. Nat Commun 2020;11:268.
47. Liao X, Song W, Zhang X, et al. An artificial peripheral neural system based on highly stretchable and integrated multifunctional sensors. Adv Funct Mater 2021;31:2101107.
48. Bai N, Wang L, Xue Y, et al. Graded interlocks for iontronic pressure sensors with high sensitivity and high linearity over a broad range. ACS Nano 2022;16:4338-47.
49. Zhu P, Du H, Hou X, et al. Skin-electrode iontronic interface for mechanosensing. Nat Commun 2021;12:4731.
50. Curry EJ, Ke K, Chorsi MT, et al. Biodegradable piezoelectric force sensor. Proc Natl Acad Sci USA 2018;115:909-14.
51. Rao J, Chen Z, Zhao D, et al. Tactile electronic skin to simultaneously detect and distinguish between temperature and pressure based on a triboelectric nanogenerator. Nano Energy 2020;75:105073.
52. Bae B, Flachsbart BR, Park K, Shannon MA. Design optimization of a piezoresistive pressure sensor considering the output signal-to-noise ratio. J Micromech Microeng 2004;14:1597-607.
53. Hu X, Chen A, Luo Y, Zhang C, Zhang E. Steerable catheters for minimally invasive surgery: a review and future directions. Comput Assist Surg (Abingdon) 2018;23:21-41.
54. Kozikowska M, Grusza M, Mrugacz G, Gagan J, Zbucka-Krętowska M, Grygoruk C. The influence of intrauterine pressure on embryo retention in a catheter after embryo transfer. Sci Rep 2019;9:11969.
55. Xingbang L, Liyun H, Lijun G. Current situation and future of coronary microvascular disease evaluation technology.
56. Meena KV, Sankar AR. Biomedical catheters with integrated miniature piezoresistive pressure sensors: a review. IEEE Sensors J 2021;21:10241-90.
57. Polygerinos P, Zbyszewski D, Schaeffter T, Razavi R, Seneviratne LD, Althoefer K. MRI-compatible fiber-optic force sensors for catheterization procedures. IEEE Sensors J 2010;10:1598-608.
58. Tan R, Benharash P, Schulam P, Schmidt JJ. Implantable electrolyte conductance-based pressure sensing catheter, II. Device construction and testing. Biomed Microdevices 2013;15:1035-41.
59. Hasenkamp W, Forchelet D, Pataky K, et al. Polyimide/SU-8 catheter-tip MEMS gauge pressure sensor. Biomed Microdevices 2012;14:819-28.
60. Huang C-H, Shih C-H, An N-J. Tactile sensor for cardiovascular catheters. In proceedings of the 3rd international conference on biomedical and bioinformatics engineering - ICBBE 16; November 2016; pp. 7-11.
61. Pandya HJ, Sheng J, Desai JP. MEMS-Based Flexible Force Sensor for Tri-Axial Catheter Contact Force Measurement. J Microelectromech Syst 2017;26:264-72.
62. Guo J, Guo S, Wang P, Wei W, Wang Y. A novel type of catheter sidewall tactile sensor array for vascular interventional surgery. In proceedings of the 2013 ICME international conference on complex medical engineering, 25-28 May 2013, Beijing, China; pp. 264-7.
63. Ahmadi M, Zhang Y, Rajamani R, Timm G, Sezen AS. A Super-capacitive pressure sensor for a urethral catheter. Annu Int Conf IEEE Eng Med Biol Soc 2018;2018:1-3.
Cite This Article
Export citation file: BibTeX | RIS
OAE Style
Li Y, Wang P, Meng C, Chen W, Zhang L, Guo S. A brief review of miniature flexible and soft tactile sensors for interventional catheter applications. Soft Sci 2022;2:6. http://dx.doi.org/10.20517/ss.2022.05
AMA Style
Li Y, Wang P, Meng C, Chen W, Zhang L, Guo S. A brief review of miniature flexible and soft tactile sensors for interventional catheter applications. Soft Science. 2022; 2(2): 6. http://dx.doi.org/10.20517/ss.2022.05
Chicago/Turabian Style
Li, Yurui, Peng Wang, Chuizhou Meng, Wenqiang Chen, Longyuan Zhang, Shijie Guo. 2022. "A brief review of miniature flexible and soft tactile sensors for interventional catheter applications" Soft Science. 2, no.2: 6. http://dx.doi.org/10.20517/ss.2022.05
ACS Style
Li, Y.; Wang P.; Meng C.; Chen W.; Zhang L.; Guo S. A brief review of miniature flexible and soft tactile sensors for interventional catheter applications. Soft. Sci. 2022, 2, 6. http://dx.doi.org/10.20517/ss.2022.05
About This Article
Copyright
Data & Comments
Data
 Cite This Article 31 clicks
Cite This Article 31 clicks













Comments
Comments must be written in English. Spam, offensive content, impersonation, and private information will not be permitted. If any comment is reported and identified as inappropriate content by OAE staff, the comment will be removed without notice. If you have any queries or need any help, please contact us at support@oaepublish.com.