Fabrication method and performance of a light-responsive hydrogel microvalve in a microfluidic chip
Abstract
Microfluidic technology has potential advantages in the complex manipulation of microfluidics on small-sized chips. However, it is difficult to integrate microvalves with complex flow channel structures, and this has limited the miniaturization of microfluidic systems and their portable applications. Light-responsive hydrogel (LRH) materials can rapidly change their volume under laser irradiation and can be used to prepare flexible microvalves to realize the integrated control of microfluidics. A simple fabrication method for an LRH microvalve on a microfluidic chip is proposed. The microspheres, as control elements of the microvalve based on an LRH modified with Laponite RD nanoclay and ferriferous oxide (Fe3O4) nanoparticles, are prepared through a T-shaped flow channel. The microvalve is assembled on the microfluidic chip with a normally closed circulation channel. The open/close performance of the microvalve is represented by the color change of the photonic crystal material. The results show that the LRH microspheres shrink and the flow channel opens after laser irradiation for 2 s. After stopping the laser at 18 s, the valve core swells and the flow channel closes.
Keywords
INTRODUCTION
Currently, microfluidic technology can realize the complex operations of microfluidics on small-sized chips. The main components of a microfluidic system are three parts, namely, the microvalve, micropump and microfluidic flow sensor[1]. Among these parts, the microvalve is the most important control element for the automation and large-scale integration of the microfluidic system[2].
According to the different driving forms, the existing traditional microvalves, including pneumatic[3,4], mechanical[5-7] and electromagnetic valves[8-10], have complex structures and numerous components, which make it difficult to fabricate microfluidic chips and limit their portability. To solve these problems, some researchers have reported microvalves based on responsive hydrogels, which can change their volume in response to changes in light[11,12], magnetic[13,14] and temperature fields[15], and pH[16,17]. The use of such materials to fabricate microvalves is expected to simplify the process of compositing microvalves in microfluidic chips and removing additional control structures to achieve high performance and low cost.
Compared with other forms of responsive hydrogel microvalves, Light-responsive hydrogel (LRH) microvalves have the advantages of fast response and simple control. Since 2004, the reports of LRH materials have increased rapidly[18]. Doping photothermal materials (such as iron oxide[19], gold[20,21], silver[22] and graphene oxide[23]) inside poly-N-isopropylacrylamide (PNIPAM) is currently a common method for preparing LRH microvalves.
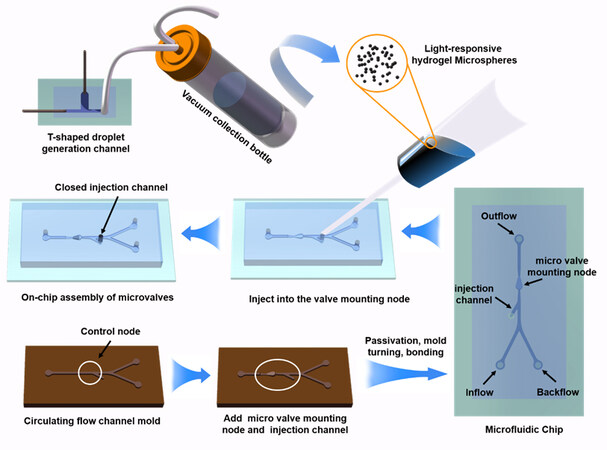
Researchers have improved the open/close speed of microvalves by modifying hydrogel materials[24,25] and selecting more efficient photothermal materials[26]. Nevertheless, few details have been reported regarding microfluidic chip designs with LRH microvalve structures and the fast characterization methods of their microfluidic control properties. With the aim of fabricating microfluidic chips by the soft lithography process[27], in this study, we propose a method to rapidly prepare an LRH microvalve in the flow channel structure of a microfluidic chip. First, the hydrogel microspheres are synthesized outside the chip, which are then injected into the microvalve mounting node on the microfluidic chip through the injection channel to complete the on-chip assembly of the microvalve. Laponite RD-modified hydrogel microspheres are prepared through a T-shaped flow channel, and the microspheres could correspondingly hybridize more ferriferous oxide (Fe3O4) nanoparticles with better photothermal conversion properties. Moreover, Fe3O4 is low-cost and easy to obtain. The microvalve structure is compounded on a microfluidic chip with a circulation channel by the above method. To characterize the liquid supply properties of microfluidic chips, it is combined with a photonic crystal film to construct a duality color-changing unit, and the surface wetting area (color-changing area) is used to rapidly and indirectly show the microfluidic liquid supply volume.
EXPERIMENTAL
Fabrication of microfluidic chip
The construction of the circulating flow channel microfluidic chip is shown in Figure 1. Both the width and height of the circulating flow channel and injection channel are 500 μm. The diameter and height of the microvalve mounting node are 1000 and 800 μm, respectively. The mold was printed out with an inkjet 3D printer (Eden 260 vs. Objet, USA). A coating machine (PDS-2010, Specialty Coating Systems, USA) was applied to evaporate a layer of Parylene film on the surface of the formed mold to ensure the subsequent curing of Polydimethylsiloxane (PDMS) and the smooth release of the formed structure. Subsequently, the polydimethylsiloxane matrix (Dow Corning, Midland, USA) and the curing agent were poured into the mold for a 4 h cure at 60 °C. After curing, the molded PDMS flow channel was peeled off. The surfaces of the cured PDMS flow channel structure and glass slide were treated by a plasma cleaner (YZD08-2C, SAOT, China), which were then bonded at 95 °C for 15 min to the construction of a microfluidic chip with the microvalve mounting node.
Preparation of LRH microspheres
According to the spherical hydrogel microspheres fabricated by this method[28], as shown in Supplementary Figure 1, the size of the LRH microspheres can be controlled by adjusting the size of the injection ports and the exit flow rate of the T-shaped flow channel. The detailed information on the specific preparation process of microspheres can be referred to in Supplementary Table 1. The pre-gel solution passing through the injection port was configured according to Table 1.
LRH configurations
| Type | NIPAM (g) | 20 nm Fe3O4 (g) | 10% w/v BIS (mL) | Solvent | 10% w/v APS (mL) |
| N01 | 3.6 | 0.4 | 5.8 | 3.68 mL water | 3.2 |
| N02 | 3.6 | 0.4 | 5.8 | 2 wt.% Lap-RD solution | 3.2 |
Characterization of LRH microspheres
Characterization of basic physical properties of microspheres
The cubic hydrogel material was prepared to characterize the swelling and deswelling properties. A volume of 80 μL of TEMED was added to the pre-gel solutions to prepare the LRH in 50 mL centrifugal tubes. After the hydrogel material reached the swelling equilibrium at room temperature, it was cut into cubes with a side length of 10 mm. A texture analyzer (TA, XT Plus, Stable) was used to test the compression resistance of the hydrogels at a compression speed of 10 mm/min until they broke.
When reaching the swelling equilibrium at room temperature, the LRH material was pre-frozen at -80 °C for 24 h and then vacuum freeze dried at -57 °C in a lyophilizer to remove moisture without any damage to the structure. After drying, the hydrogel samples were sputtered with gold and analyzed by field-emission scanning electron microscopy (SU8010LA, Hitachi) equipped with energy dispersive spectroscopy to obtain the morphology of the LRH microspheres and the distribution of photothermal nanomaterials.
Characterization of swelling characteristics of LRH materials
The swelling characteristics of the above LRH were characterized by gravimetry and each test was repeated three times. The hydrogel sample was dried at room temperature until the weight did not change and then immersed in water at 20 °C for swelling. After 10 min, the sample was removed from the water. After wiping the surface, the sample was weighed and then put back into the water to swell. This step was repeated several times.
Furthermore, the deswelling properties of the hydrogel materials were characterized. The hydrogels at the swelling equilibrium were placed in water at 50 °C. After 10 min, the samples were taken out of the water to weigh on the wiping surface. This step was repeated several times.
Characterization of photothermal properties of LRH microspheres
A CCD camera (GS3-U3-23S6C-C, Point grey, USA) was used to collect the image information of the spherical microvalve. Image J software was used to process the graphic information to characterize the particle size of the microvalve. Ultimately, the structural parameters of the T-shaped flow channel were determined. The LRH micro-spheres were placed in a 23 mm-diameter Petri dish with the addition of 2 mL of distilled water. A NIR laser (2 W, 808 nm, Laserland) was used to irradiate the valve core with a spot diameter of 1 mm. An infrared camera (E8xt 2.1L, FLIR, Estonia) was applied to measure the two types of water condensation. The temperature of the glued sample was measured three times. The Lap-RD-modified microsphere material was heated using a water bath (HH-2, LICHEN, China) at a constant temperature in the range of 20 °C-50 °C (holding 5 min for every 1 °C) to analyze the temperature-dependent characteristics of the volume of the microspheres. Finally, the photothermal reversibility of the LRH microspheres was studied. The hydrogel was irradiated by a NIR laser for 10 s, which was then turned off to allow the hydrogel to cool to room temperature naturally. This was repeated five times for further post-processing.
The preparation of LRH microspheres was performed through a T-shaped flow channel with a CCD camera (GS3-U3-23S6C-C, Point grey, USA) to collect the image information of the microspheres. ImageJ software was used to process image information to characterize the particle size. The LRH microspheres were placed in a 23 mm-diameter Petri dish with the addition of 2 mL of distilled water. The microspheres were irradiated with a NIR laser (1 W, 808 nm, Laserland) and the diameter of the laser spot was 1 mm. The temperature of the two hydrogel samples was measured using an infrared camera (E8xt 2.1 L, FLIR, Estonia). Three measurements were taken for each sample.
On-chip assembly and performance characterization of the microvalve
The on-chip assembly process of the microvalve is shown in Figure 1. The hydrogel microsphere with a diameter of 1.7 mm was placed in distilled water at 70 °C (the volume phase transition temperature) for the rapid shrinkage of volume. The hydrogel microsphere with a sufficiently small volume was then sucked into the syringe and injected into the microvalve mounting node from the injection channel. When the temperature drops, the volume of the hydrogel microsphere expands and occupies the internal space of the microvalve structure. Finally, the
RESULTS AND DISCUSSION
Physical properties of LRH
As shown in Supplementary Figure 2, LRH microspheres with different particle sizes were prepared by a
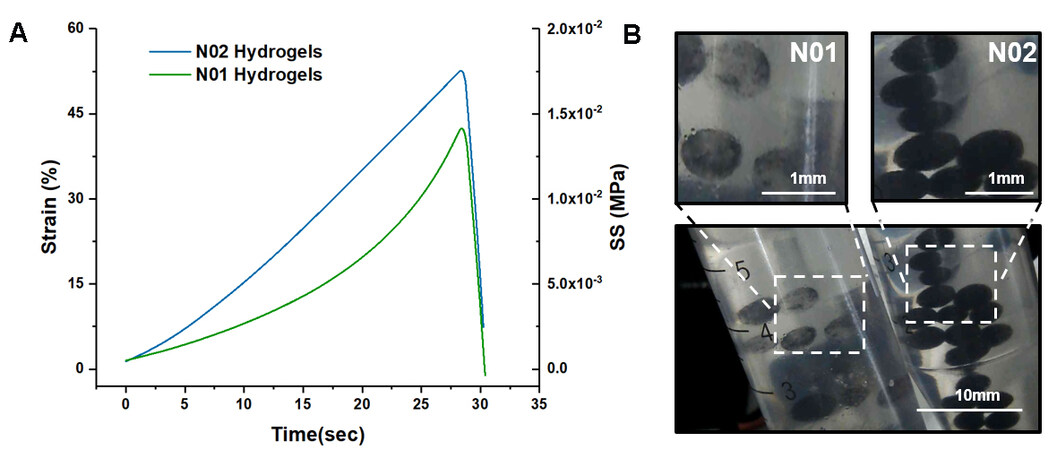
Figure 2. Characterization results of the physical properties of hydrogels. (A) Compressive strength curves of two hydrogels. (B) Photographs of “N01” and “N02” LRH microspheres. LRH: Light-responsive hydrogel.
Figure 3A and C show the SEM images of the internal morphologies of the “N01” and “N02” LRH microspheres. Both “N01” and “N02” showed a uniform porous network structure. Compared to the “N01” hydrogel without Lap-RD modification, the “N02” hydrogel had a multistage pore structure and the primary pore was significantly larger than “N01”. This was because the difference in the reactivity ratio of BIS and NIPAM led to a disordered distribution of the crosslinking points of the gel. After modification by Lap-RD, two charges were simultaneously presented on the orderly Lap-RD wafers in solution, resulting in the formation of ordered physical crosslinks of poly NIPAM molecular chains in solution. Therefore, the “N02” hydrogel could initiate the chain initiation and chain termination on the RD sheet structure during the crosslinking process, contributing to the formation of a uniform multilevel pore structure.
Figure 3. Characterization results of the physical properties of hydrogels. (A) SEM image of “N01” hydrogel. (B) EDS characterization diagram of Fe element dispersion in “N01” LRH. (C) SEM image of “N02” hydrogel. (D) EDS characterization diagram of Fe element dispersion in “N02” LRH. EDS: Energy dispersive spectroscopy; LRH: light-responsive hydrogel.
As Figure 3B shows, the Fe3O4 nanoparticles in the “N01” hydrogel generally produced the agglomeration phenomenon. With the comparison of Figure 3B and D, the distribution of Fe3O4 nanoparticles was more uniform in the Lap-RD modified “N02” hydrogel, which could be attributed to the modification effect of Lap-RD. The lamellar layer of the foil was charged negatively and the end face was charged positively, which could rapidly form a three-dimensional colloidal structure with a uniform spatial arrangement in water, therefore increasing the viscosity of the system with a high degree of suspension. Thus, the increase in suspension of the pre-gel solution made the nanosized ferrous tetrachloride photothermal material difficult to precipitate and agglomerate.
The structural difference between the two hydrogels caused the difference in mechanical properties. The above results preliminarily indicate that Lap-RD could make hydrogels obtain a richer microstructure, thereby improving the mechanical properties of traditional chemically crosslinked hydrogels. Lap-RD also improved the suspension of the pre-gel solution and made the photothermal nanomaterial more uniformly dispersed in the LRH microspheres.
Swelling properties of LRH microvalve core material
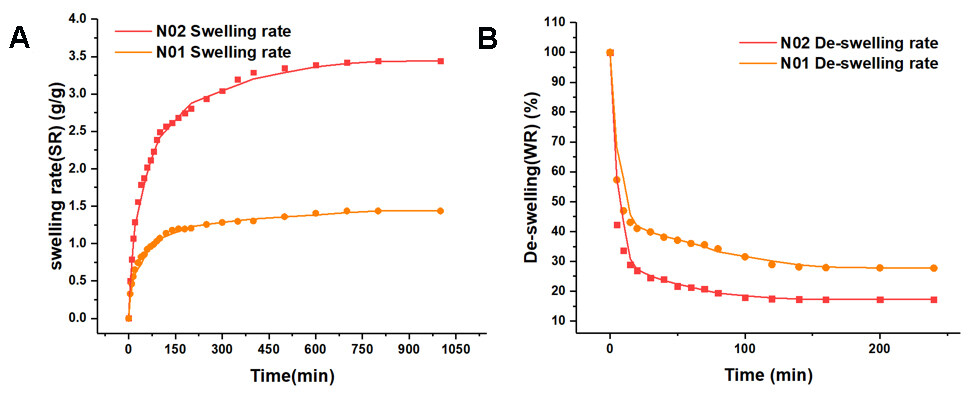
Figure 4 shows the swelling and deswelling curves of the “N01” and “N02” hydrogels at 20 °C-50 °C. Both hydrogels presented a good temperature sensitivity because the macromolecular chain extended due to hydration when the temperature was lower than the phase transition temperature. Moreover, the molecular chain also showed an extended conformation related to the swelling of hydrogels in an aqueous solution. When the temperature was higher than the phase transition temperature, the gel underwent rapid dehydration and condensation. Owing to the mutual attraction of the hydrophobic groups, the molecular chain conformation shrunk, exhibiting a deswelling phenomenon. The microvalve spool matrix is a temperature-sensitive hydrogel, which shrinks rapidly in water above the lower critical solution temperature (LCST). As seen in Figure 4A, the swelling speed of the “N02” hydrogel was faster than that of the “N01” hydrogel. According to a previous study[29], the swelling rate of the “N02” hydrogel was ~2.8 g/g at 200 min, while the “N01” hydrogel was only 1.2 g/g at 200 min. Figure 4B shows the deswelling rate of the “N01” and “N02” hydrogels at 20 °C (below LCST) to 50 °C (above LCST).
Figure 4. Comparison of swelling and deswelling properties of two kinds of hydrogels: (A) swelling rate; (B) deswelling rate.
Compared with the “N01” hydrogel, the “N02” hydrogel showed a faster shrinkage and more water loss simultaneously. After soaking in water at 50 °C for 15 min, the volume of “N02” rapidly shrunk and the water loss was nearly 80%. In contrast, “N01” only released ~60% of water. This was due to the addition of Lap-RD, which increased the pore size of the “N02” hydrogel and generated a multistage pore structure (more space was provided for the flow of feed water). With the water more easily diffusing, the swelling performance of the hydrogel material could be improved significantly. The modification of Lap-RD greatly improved the swelling and deswelling performance, meaning that the modified hydrogel valve core could achieve a faster rate of volume change. In addition, the modification of Lap-RD did not fundamentally change the volume phase transition temperature (VPTT) of PNIPAM, which only increased the swelling rate of the hydrogel.
Photothermal conversion performance of microsphere materials
The constructed on-chip LRH microvalve could be controlled by a NIR laser without any contact. The Fe3O4 photothermal nanomaterial dropping in the microvalve core absorbed infrared light and generated heat. The total energy generated was associated with the distance, number, and arrangement of the photothermal nanoparticles. For the same type of samples, qualitative analyses of their photothermal conversion performance were carried out through the infrared thermometerter. The “N01” and “N02” hydrogel microspheres with a particle size of 1.7 mm were tested. Figure 5A shows the temperature change of an LRH microsphere immersed in water irradiated by a 1 W/cm2 NIR laser with a wavelength of 808 nm. The LRH microspheres rapidly absorbed energy, which heated up under the irradiation of the NIR light. The temperature change process of the hydrogel microspheres was observed by an infrared camera, and the highest temperature of its center was measured to investigate the photothermal conversion performance of the two hydrogels.
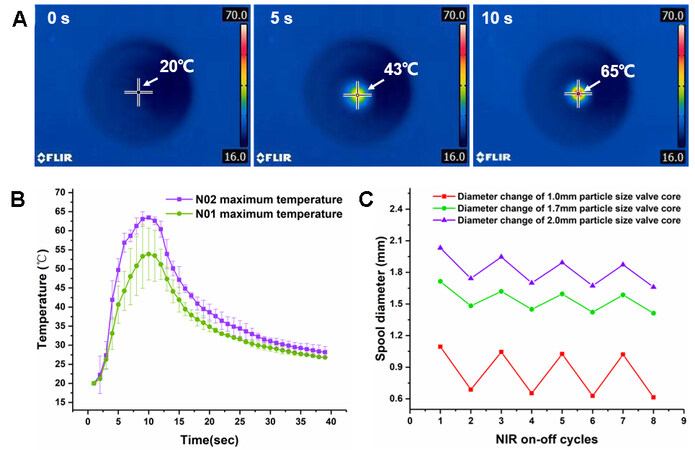
Figure 5. Analysis of the photothermal performance of hydrogels. (A) Heating process of N02 hydrogel recorded by an infrared camera. (B) Heating curve of two hydrogel materials. (C) Particle size changes of N02 hydrogel with different particle sizes under cyclic laser irradiation.
As shown in Figure 5B, the microspheres prepared by the “N02” hydrogel were all heated to ~65 °C after 10 s of laser irradiation, indicating a good photothermal consistency. The temperature changing trend of unmodified “N01” hydrogel microspheres was similar to that of “N02”. They both reached the highest temperature before stopping the laser irradiating. However, the highest heating temperatures of the same batch of “N01” hydrogel microspheres differed greatly. Generally, the “N02” hydrogel modified with Lap-RD had an obvious photothermal conversion and stability compared with the “N01” hydrogel when exposed to the 808 nm NIR laser. The experimental results could be analyzed by combining the dispersion of the photothermal materials in the two hydrogels. The Fe3O4 particles were easily agglomerated and precipitated in the “N01” pre-gel solution. During the preparation of hydrogel microspheres by the
The stability and repeatability of the swell-shrink transition of the Lap-RD-modified hydrogel microspheres were also studied. As shown in Figure 5C, three different particle sizes of “N02” photoresponsive hydrogel microspheres were exposed to five cycles of NIR laser irradiation. The particle sizes of the three microspheres basically remained unchanged and the volume reduction of the hydrogel microspheres was less than 3%. These results indicate that the light response volume change of this kind of microvalve was reversible and repeatable.
Performance and characterization of microvalve for microfluidic chip
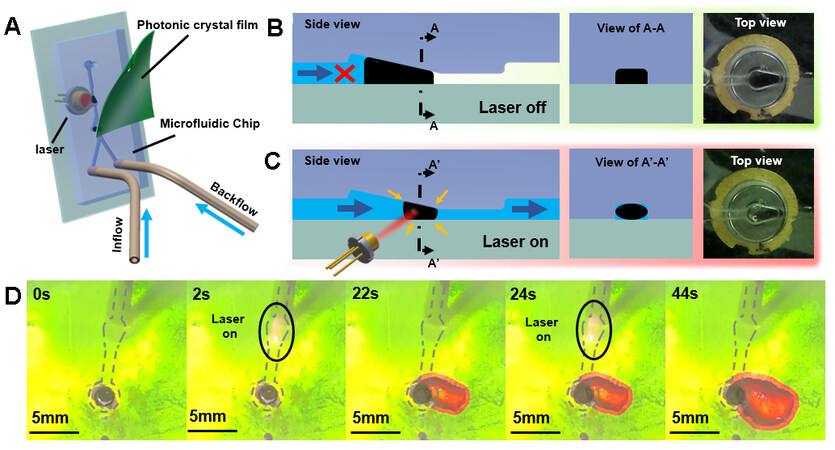
The mechanical properties and photothermal conversion properties of the LRH materials were improved by modifying the traditional NIPAM hydrogel with Lap-RD materials. The surface of the photonic crystal film was subjected to hydrophilic treatment, which was then attached to the upper layer of the circulating flow channel assembled with the microvalve [Figure 6A]. As observed in the color change of the photonic crystal layer, the time required for turning on and off can be characterized. Deionized water was introduced from the inlet of the microfluidic chip at a fixed flow rate of 5 mL/min. At this time, no color change was observed on the surface of the photonic crystal film, meaning that there was no liquid flowing out from the main outlet.
Figure 6. Characterization of the performance of LRH microvalves. (A) Schematic diagram of duality color-changing unit assembly. (B) Hydrogel microvalve in the closed state; from left to right are the side view, A-A cross-sectional view and the actual top view. (C) Hydrogel microvalve in the open state; from left to right are the side view, A’-A’ cross-section Schematic diagram and physical top view. (D) Color change process of photonic crystal layer on the surface of the duality color-changing unit after hydrogel microvalve is opened (dotted line indicates the position of the flow channel under the film, the green area is the initial color of the photonic crystal film and the red area indicates that the film is wetted by water). LRH: Light-responsive hydrogel.
As shown in Figure 6B and D, the LRH microvalve was in a closed state at 0 s with a better sealing performance at the flow rate of 10 mL/min. When the laser was switched on, the microvalve opened with a 1 W and 808 nm NIR light for 2 s. As shown in Figure 6C, the volume of the hydrogel microspheres in the microvalve shrunk, and the water could pass through the gap created by the shrinkage of the microspheres. In this state, the microvalve was opened and the liquid flowed out from the main outlet to wet the surface of the photonic crystal film. As shown in Figure 6D, the solute among the photonic crystal structures was replaced by water from the air after the film was wetted and the color of the part wetted by water changed from the original green to red. After stopping the laser irradiation, the liquid continuously flowed through the hydrogel microspheres, which would take away heat. When the temperature of the hydrogel microspheres was lower than the VPTT, the hydrogel microspheres began to absorb water and swell. After 18 s, the microspheres reoccupied the internal space of the microvalve, making it close again. The discoloration area of the photonic crystal layer stopped changing at this time, showing that the opening and closing response time of the microvalve was ~20 s.
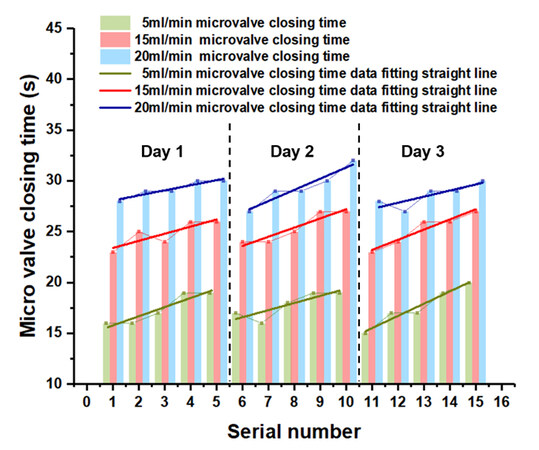
The reproducibility of the hydrogel microvalve was studied at different flow rates. When the inlet flow rate was lower than 1 mL/min, the water flow could not pass through the narrow slits of the hydrogel microspheres and the microvalve was closed. When the inlet flow rate was greater than 20 mL/min, the liquid leakage would occur at the microvalve. Therefore, the effective working range of flow rate in the microvalve structure was 1-20 mL/min. As shown in Figure 7, the period from the opening state to the closing state of the microvalve became longer as the flow rate increased. During the five consecutive tests on the same day, the closing time of the microvalve gradually increased. After a full day of swelling, the closing time of the microvalve returned to the initial level. However, the subsequent continuous laser irradiation would prolong the closing time of the microvalve. These experimental results indicated that the valve actuation was reversible and the LRH microvalve had the potential for achieving flow control in microfluidic chips. The microvalve would still work properly when it carried out opening and closing cycles 20 times a day for 5 d. However, if the laser was shone continuously for 10 s or more, the microvalve would be destroyed.
CONCLUSIONS
This study modified a temperature-sensitive hydrogel material using the Laponite RD matrix material to enhance the swelling capacity and mechanical properties of the original material. The modification of RD also made the Fe3O4 nanoparticles distribute uniformly, contributing to the consistent light-to-heat conversion performance of the hydrogel material. LRH microspheres were prepared using a T-shaped flow channel, which was controlled by a single outlet. Based on the circulating flow channel microfluidic chip, the microspheres were assembled on the chip through the injection channel to complete the on-chip construction of the microvalve. The duality color-changing unit was constructed according to the photochromic mechanism of photonic crystal, and the open/close performance of microvalve was characterized by the color change of the photonic crystal. The experimental results showed that the LRH microvalve had a good opening and closing response. The fluid would open after the microspheres were irradiated by laser for 2 s. Meanwhile, the fluid flowing through the microspheres would take away the heat. After the microspheres absorbed water and swelled for ~18 s, the fluid closed. The microvalve prepared in this study had a good open/close performance and stable repeatability.
DECLARATIONS
AcknowledgmentsThanks for the preliminary exploration work done by Shuai Wang for this study. Thanks for Shuzhang Liang’s advice on this study. Thanks for Nan Zhao’s suggestion on the English grammar of this article.
Authors’ contributionsMade substantial contributions to conception and design of the study and performed data analysis and interpretation: Zhang W
Performed data acquisition: Pan J, Chen K
Provided administrative and technical support: Zhang D, Feng L
Availability of data and materialsNot applicable.
Financial support and sponsorshipThis work was supported by The National Science Foundation Program of China (51975574) and The Fundamental Research Funds for the Central Universities(2020TC017).
Conflicts of interestAll authors declared that there are no conflicts of interest.
Ethical approval and consent to participateNot applicable.
Consent for publicationNot applicable.
Copyright© The Author(s) 2022.
Supplementary MaterialsREFERENCES
1. Qian JY, Hou CW, Li XJ, Jin ZJ. Actuation mechanism of microvalves: a review. Micromachines (Basel) 2020;11:172.
3. Goldowsky J, Knapp HF. Gas penetration through pneumatically driven PDMS micro valves. RSC Adv 2013;3:17968.
4. Perdigones F, Luque A, Quero J. Pneumatically actuated positive gain microvalve with n-channel metal-oxide semiconductor-like behaviour. Micro Nano Lett 2011;6:363.
5. Ferraro D, Serra M, Ferrante I, Viovy J, Descroix S. Microfluidic valve with zero dead volume and negligible back-flow for droplets handling. Sensors and Actuators B: Chemical 2018;258:1051-9.
6. Guler MT, Beyazkilic P, Elbuken C. A versatile plug microvalve for microfluidic applications. Sensors and Actuators A: Physical 2017;265:224-30.
7. Kaminaga M, Ishida T, Omata T. Fabrication of pneumatic microvalve for tall microchannel using inclined lithography. Micromachines (Basel) 2016;7:224.
8. Chang PJ, Chang FW, Yuen MC, Otillar R, Horsley DA. Force measurements of a magnetic micro actuator proposed for a microvalve array. J Micromech Microeng 2014;24:034005.
9. Gholizadeh A, Javanmard M. Magnetically actuated microfluidic transistors: miniaturized micro-valves using magnetorheological fluids integrated with elastomeric membranes. J Microelectromech Syst 2016;25:922-8.
10. Liu TG, Wu J, Xia C, Qian ZH. A Microvalve driven by a ferrofluid-based actuator. AMR 2012;433-440:3767-72.
11. Fan P, Fan Z, Huang F, et al. GO@polyaniline nanorod array hierarchical structure: a photothermal agent with high photothermal conversion efficiency for fast near-infrared responsive hydrogels. Ind Eng Chem Res 2019;58:3893-901.
12. Lee E, Lee H, Yoo SI, Yoon J. Photothermally triggered fast responding hydrogels incorporating a hydrophobic moiety for light-controlled microvalves. ACS Appl Mater Interfaces 2014;6:16949-55.
13. Ghosh S, Yang C, Cai T, Hu Z, Neogi A. Oscillating magnetic field-actuated microvalves for micro- and nanofluidics. J Phys D: Appl Phys 2009;42:135501.
14. Satarkar NS, Zhang W, Eitel RE, Hilt JZ. Magnetic hydrogel nanocomposites as remote controlled microfluidic valves. Lab Chip 2009;9:1773-9.
15. Baldi A, Lei M, Gu Y, Siegel RA, Ziaie B. A microstructured silicon membrane with entrapped hydrogels for environmentally sensitive fluid gating. Sensors and Actuators B: Chemical 2006;114:9-18.
16. Beebe DJ, Moore JS, Bauer JM, et al. Functional hydrogel structures for autonomous flow control inside microfluidic channels. Nature 2000;404:588-90.
17. Yu Q, Bauer JM, Moore JS, Beebe DJ. Responsive biomimetic hydrogel valve for microfluidics. Appl Phys Lett 2001;78:2589-91.
18. Schiphorst J, Saez J, Diamond D, Benito-Lopez F, Schenning APHJ. Light-responsive polymers for microfluidic applications. Lab Chip 2018;18:699-709.
19. Yoon J, Bian P, Kim J, McCarthy TJ, Hayward RC. Local switching of chemical patterns through light-triggered unfolding of creased hydrogel surfaces. Angew Chem Int Ed Engl 2012;51:7146-9.
20. Wei Q, Ji J, Shen J. Synthesis of near-infrared responsive gold Nanorod/PNIPAAm Core/Shell Nanohybrids via surface initiated ATRP for smart drug delivery. Macromol Rapid Commun 2008;29:645-50.
21. Sershen SR, Mensing GA, Ng M, Halas NJ, Beebe DJ, West JL. Independent optical control of microfluidic valves formed from optomechanically responsive Nanocomposite hydrogels. Adv Mater 2005;17:1366-8.
22. Liu Y, Liu X, Yang J, Lin D, Chen X, Zha L. Investigation of Ag nanoparticles loading temperature responsive hybrid microgels and their temperature controlled catalytic activity. Colloids and Surfaces A: Physicochemical and Engineering Aspects 2012;393:105-10.
23. Shen J, Yan B, Li T, Long Y, Li N, Ye M. Mechanical, thermal and swelling properties of poly(acrylic acid)-graphene oxide composite hydrogels. Soft Matter 2012;8:1831-6.
24. Schiphorst J, Coleman S, Stumpel JE, Ben Azouz A, Diamond D, Schenning APHJ. Molecular design of light-responsive hydrogels, for in situ generation of fast and reversible valves for microfluidic applications. Chem Mater 2015;27:5925-31.
25. Satoh T, Sumaru K, Takagi T, Takai K, Kanamori T. Isomerization of spirobenzopyrans bearing electron-donating and electron-withdrawing groups in acidic aqueous solutions. Phys Chem Chem Phys 2011;13:7322-9.
26. Jadhav AD, Yan B, Luo RC, et al. Photoresponsive microvalve for remote actuation and flow control in microfluidic devices. Biomicrofluidics 2015;9:034114.
27. Qin D, Xia Y, Whitesides GM. Soft lithography for micro- and nanoscale patterning. Nat Protoc 2010;5:491-502.
28. Luo RC, Ranjan S, Zhang Y, Chen CH. Near-infrared photothermal activation of microgels incorporating polypyrrole nanotransducers through droplet microfluidics. Chem Commun (Camb) 2013;49:7887-9.
Cite This Article
Export citation file: BibTeX | RIS
OAE Style
Pan J, Chen K, Zhang W, Feng L, Zhang D. Fabrication method and performance of a light-responsive hydrogel microvalve in a microfluidic chip. Soft Sci 2022;2:4. http://dx.doi.org/10.20517/ss.2022.03
AMA Style
Pan J, Chen K, Zhang W, Feng L, Zhang D. Fabrication method and performance of a light-responsive hydrogel microvalve in a microfluidic chip. Soft Science. 2022; 2(2): 4. http://dx.doi.org/10.20517/ss.2022.03
Chicago/Turabian Style
Pan, Jingwen, Kehan Chen, Wenqiang Zhang, Lin Feng, Deyuan Zhang. 2022. "Fabrication method and performance of a light-responsive hydrogel microvalve in a microfluidic chip" Soft Science. 2, no.2: 4. http://dx.doi.org/10.20517/ss.2022.03
ACS Style
Pan, J.; Chen K.; Zhang W.; Feng L.; Zhang D. Fabrication method and performance of a light-responsive hydrogel microvalve in a microfluidic chip. Soft. Sci. 2022, 2, 4. http://dx.doi.org/10.20517/ss.2022.03
About This Article
Copyright
Data & Comments
Data
 Cite This Article 17 clicks
Cite This Article 17 clicks














Comments
Comments must be written in English. Spam, offensive content, impersonation, and private information will not be permitted. If any comment is reported and identified as inappropriate content by OAE staff, the comment will be removed without notice. If you have any queries or need any help, please contact us at support@oaepublish.com.