Simultaneously enhancing moisture and mechanical stability of flexible perovskite solar cells via a polyimide interfacial layer
Abstract
Perovskite solar cells (PSCs) have aroused tremendous attention due to the high power conversion efficiency (PCE) and flexibility of the organic-inorganic hybrid perovskite films. However, the commercialization of perovskite solar cells is still impeded due to the instability issue induced by moisture and mechanical stress. Herein, we introduce soluble hydrophobic polyimide (PI) as an interfacial layer on top of the perovskite film to block the infiltration of moisture into the perovskite film. The MAPbI3-based solar cell with the insertion of PI layer exhibited an impressive stability, maintaining 87% of the initial PCE even after exposing to 50% relative humidity for 550 h and presenting a decent PCE of 21.22% due to its ability to extract holes and reduce trap-assisted recombination. Moreover, the high tolerance of PI to the mechanical stress gives a more stable flexibility to the PSCs under constant bending.
Keywords
INTRODUCTION
Organic-inorganic hybrid metal halide perovskite (OMHP) materials, as one of the most promising light-harvesting materials for photovoltaic devices, have been widely applied in perovskite solar cells (PSCs) by their virtues of outstanding optoelectronic properties and easy fabricating process[1-3]. Further, the flexibility of PSCs can help their integration with other power sources in wearable electronics[4]. To date, the certified power conversion efficiency (PCE) has reached 25.5%[5-7], steadily approaching the performance of monocrystalline silicon solar cells. Nevertheless, the instabilities of OMHP caused by heat, oxygen, moisture, or mechanical stress tremendously hinder the commercialization of PSCs[8-10].
Considering the ionic interaction in OMHP, water is the dominating factor during the degradation process at ambient atmosphere. As for methylammonium lead triiodide (MAPbI3), its exposure to moisture would quickly and reversibly transform the original perovskite phase into intermediate monohydrates or even dihydrates byproducts, which would further irreversibly decompose into CH3NH3I and PbI2, as illustrated in Equations (1) and (2)[11].
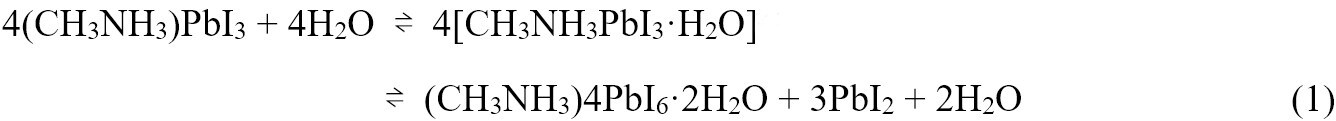
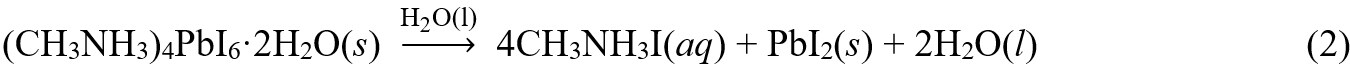
To prevent the perovskite from being destroyed by moisture, a possible solution is to utilize hydrophobic hole-transporting materials (HTMs). For example, Xie et al.[12] introduced the hydrophobic and dopant-free Co-Ni bimetal-doped carbon aerogels simultaneously as HTM and electrode, remarkably improving the long-term stability of perovskite: 86% of the initial efficiency was maintained even after 1296 h exposure in air. Similarly, Zheng et al.[13] and Li et al.[14] adopted the hydrophobic 2D perovskite materials as HTM and thus enhanced the moisture resistance of perovskite. Despite their excellent moisture stability, these stable devices always suffered from low efficiencies compared with the devices based on spiro-OMeTAD HTM. Instead, when a small amount of 2D perovskite is added into 3D perovskite, both moisture stability and photoelectric performance can be improved[15,16].
Another important strategy is to apply effective water barrier polymer layers to encapsulate the perovskite film. Kim et al.[17] demonstrated that hygroscopic polyethylene oxide can absorb water before the perovskite layer, preventing water from permeating into the perovskite film. On the other hand, a hydrophobic polymer layer can also protect perovskite from water penetration. Zhang’s group deposited polystyrene on top of the perovskite layer, maintaining 73% of the initial PCE after 60 h in 80% humidity[18]. These approaches show the feasibility of employing a polymer as a functional layer to enhance the stability of perovskite films. Polyimide (PI) is a kind of multifunctional engineering materials with a high tolerance to temperature and mechanical stress. PI has been used as an additive to enhance the photovoltaic performance of PSCs[19] and as foldable substrates in flexible devices[20,21]. Nevertheless, few studies apply PI as an interfacial layer.
We introduced a soluble hydrophobic polymer, PI, by a spin-coating deposition method on top of perovskite films. By employing PI as the interfacial layer, a PCE of 21.22% was achieved based on MAPbI3, which could maintain 87% of the original PCE after more than 550 h aging in 50% relative humidity (RH) and 72% of the original PCE after 2500 cycles of bending. Further detailed studies showed that PI layer not only exhibits an excellent ability in extracting holes but also passivates the surface traps.
EXPERIMENTAL
Material and solvent
Methylammonium iodide (MAI, 99.5%) was purchased from Xi’an Polymer Light Technology Corp. Lead iodide (PbI2, 99.999%) and 2,2',7,7'-tetrakis(N,N-di-p-methoxyphenylamine)-9,9-spirobifluorene (spiro-OMeTAD, 99.8%) were purchased from Yingkou Libra New Energy Technology Co., Ltd. The aqueous dispersion of PEDOT:PSS (AI 4083) was obtained from Shanghai MaterWin New Materials Co., Ltd. Bis(trifluoromethanesulfonyl)imide (Li-TFSI, 99.5%), tert-butylpyridine (t-BP, 98%), and Tin(II) chloride dihydrate (SnCl2·2H2O, 98%) were purchased from Sigma-Aldrich. A soluble polyimide (PI, 5 wt% in N-Methylpyrrolidone) was obtained from ShenZhen Dalton Electronic Material Co., Ltd. As for solvent, dimethyl formamide (DMF, 99.8%), dimethyl sulfoxide (DMSO, 99.9%), chlorobenzene (CB, 99.8%), acetonitrile (99.8%), and butanol (99.8%) were purchased from Sigma-Aldrich and used without further purification. FTO glasses (FTO, 7 Ω per square) were purchased from Advanced Election Technology Co., Ltd.
Solution preparation
The SnO2 nanocrystalline (NC) solution was prepared by our previous method[22]. Typically, the SnCl2·2H2O solution (0.1 M) was prepared by dissolving SnCl2·2H2O separately in 20 mL butanol containing 2 mL water in a flask. Then, the solution was separately stirred at room temperature for 10 min. To synthesize colloidal SnO2 NC, the SnCl2·2H2O solution was refluxed at 110 °C for 2-4 h with an open refluxing apparatus.
The perovskite precursor was prepared by dissolving 922 mg PbI2 and 318 mg MAI into 1.6 mL mixed solvent of DMF and DMSO (7:3 v/v). Spiro-OMeTAD solution was prepared by dissolving 72 mg spiro-OMeTAD into 1 mL CB, with the dopant of 17.5 μL Li-TFSI solution (520 mg in 1 mL acetonitrile) and
Perovskite solar cells fabrication
The FTO substrates were sequentially ultrasonically cleaned with detergent, deionized water, and isopropanol for 15 min, respectively. Before spin-coating SnO2 ETLs, the FTO substrates were further cleaned by UV-ozone for 15 min. Then, the SnO2 NC solution was dropped on the FTO substrates and spin-coated at 500 rpm for 3 s and 3000 rpm for 30 s, followed by thermal annealing at 150 °C for 1 h. After the substrates cooled down, a 15 min UV-ozone treatment was operated again. Then, all substrates were transfer into a glovebox filled with N2 atmosphere to complete the subsequent process. The perovskite precursor solution was spin-coated on SnO2 layer at 500 rpm for 3 s and 4000 rpm for 30 s. Then, 400 μL CB as antisolvent were dripped onto the perovskite film 21 s prior to the end of the second spinning program. Afterward, the perovskite film was annealed on hotplate at 100 °C for 10 min. PI solution (0.5, 1.0, 1.5, and 2.0 wt% in CB) was spin-coated at 500 rpm for 3 s and 4000 rpm for 30 s, followed by 20 min annealing treatment at 100 °C. When it cooled down, spiro-OMeTAD solution was spin-coated upon the perovskite film at 500 rpm for 3 s and 3000 rpm for 30 s. Finally, the Ag electrode was thermally evaporated on top of the device under high vacuum condition (< 5 × 10-4 Pa). The active area of all devices was 0.07 cm2 defined by a metal mask.
Characterization
The surface and cross-section morphologies of perovskite and PSCs devices were investigated by FE-SEM (ZEISS Ultra-55). The ultraviolet and visible spectrophotometry (UV-vis) absorption spectra of perovskite film were measured by SHIMADZU UV-2700 in the wavelength range of 450-850 nm. The surface roughness of the perovskite film was obtained from atomic force microscope (AFM) (Asylum Research, Cypher). The water contact angle of the perovskite surface was measured by contact angle tester (Dataphysics OCA20, German). Ultraviolet photoelectron spectroscopy was performed on SHIMADZU AXIS SUPRA. The Fourier transform infrared (FTIR) spectrum was conducted on Bruker Vertex 70. The current-voltage (J-V) curves of the non-encapsulated solar cells were measured by Keithley 2400 by forward scan (1.2 to -0.2 V) with a scanning rate of 50 mV/s (voltage step of 10 mV and delay time of 50 ms) in a glovebox under AM 1.5 G illuminations (100 mW·cm-2) from a solar simulator (Newport, 91160), which was calibrated using a standard silicon solar cell device by the NREL. Steady-state photoluminescence (PL) spectra were measured under an excitation wavelength of 460 nm by using Lengguang F97pro Fluorescence Spectrophotometer. Time-resolved photoluminescence (TRPL) spectra were recorded by a spectrofluorometer (Edinburgh FLS9800) under a 450 nm excitation. Electrochemical impedance spectroscopy (EIS) was studied on an electrochemical workstation (CHI660) from 1 MHz to 1 Hz, biased at 0.2 V in the dark. X-ray diffraction (XRD) (PANalytical X’Pert PRO) was performed with Cu Kα radiation under operating conditions of 40 kV and 40 mA. The space charge limited current (SCLC) characteristic of the devices was measured using a Keithley 2400 in the dark. According to SCLC theory, the defect (trap) density (ntrap) can be estimated as follows: ntrap = (2εε0VTFL)/(eL2), where L, ε, ε0, and e are the thickness of the perovskite film (ca. 530 nm), dielectric constant of the material, permittivity of vacuum, and electronic charge, respectively. The external quantum efficiency (EQE) measurements of devices were carried out using QE-C system (Taiwan, Enlitech) in a wavelength range of 300-900 nm.
RESULTS AND DISCUSSION
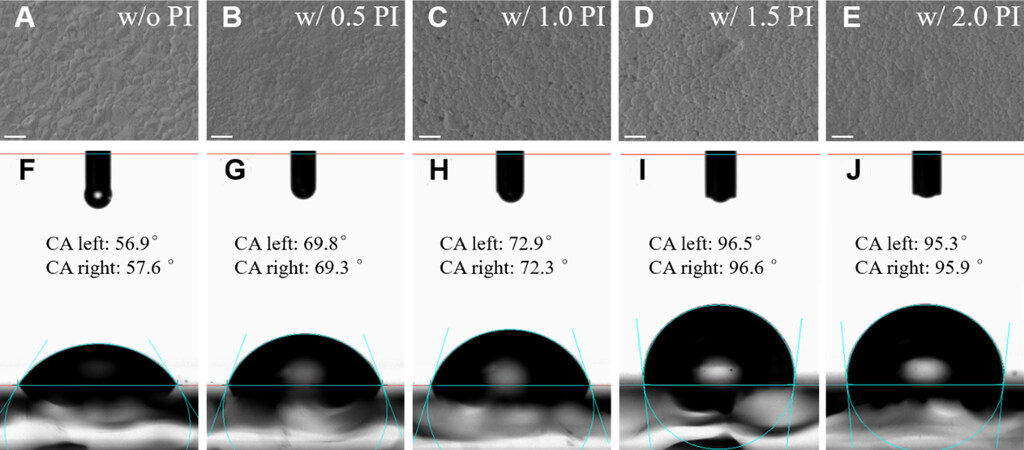
The concentration of the PI was optimized by characterizing the morphology of PI films on top of the perovskite with the top-view scanning electron microscopy (SEM), as shown in Figure 1A-E. The pristine perovskite film [Figure 1A] shows a compact surface with grain sizes ranging from 200 to 600 nm. After combining the PI layer [Figure 1B-E], the morphologies of perovskite films have no apparent changes compared with that of pristine film. Specially, PI at lower concentration (0.5 wt%) gives a relatively flat and continuous morphology, which was also confirmed by the AFM, as illustrated in Supplementary Figure 1. The water contact angles were largely changed after the inset of the PI layer on top of the perovskite films, as shown in Figure 1F-J. Due to the high hydrophobicity of PI, the contact angle of perovskite was sharply increased from 56.9° (pristine) to 95.3° (PI layer at the concentration of 2 wt%) upon the insertion of PI interfaces, providing the possibility of efficient inhibiting of moisture degradation[23]. By measuring the ultraviolet photoelectron spectroscopy curves of the perovskite films with and without PI, the energy level diagram of the PSC could be drawn [Supplementary Figure 2]. Due to the thin thickness of PI (~10 nm, as shown in Supplementary Figure 3), holes from the value band maximal of perovskite can tunnel to spiro-OMeTAD[24,25]. The FTIR spectrum of pure PI is presented in Supplementary Figure 4, showing the characteristic band peaks of the imide carbonyl were 1779 cm-1 (asymmetric C=O, stretching), 1721 cm-1 (symmetric C=O, stretching), and 1362 cm-1 (C-N, stretching)[26,27].
Figure 1. Top-view SEM images of pristine perovskite films (A). Top-view SEM images of perovskite films modified by PI solution with different concentrations: (B) 0.5 wt%; (C) 1.0 wt%; (D) 1.5 wt%; and (E) 2.0 wt%. The corresponding water contact angles based on the above perovskite films: (F) for (A); (G) for (B); (H) for (C); (I) for (D); and (J) for (E). SEM: Scanning electron microscopy. PI: polyimide.
Planar PSCs with configuration of FTO/SnO2/MAPbI3/PI/spiro-OMeTAD/Ag were fabricated, with the thickness of SnO2, MAPbI3, spiro-OMeTAD, and Ag being about 30, 300, 140, and 80 nm, respectively, as shown in the cross-sectional SEM image [Supplementary Figure 5]. These PSCs were then measured under an irradiation intensity of 100 mW/cm2 (AM 1.5 G solar spectrum), and the current density-voltage curves
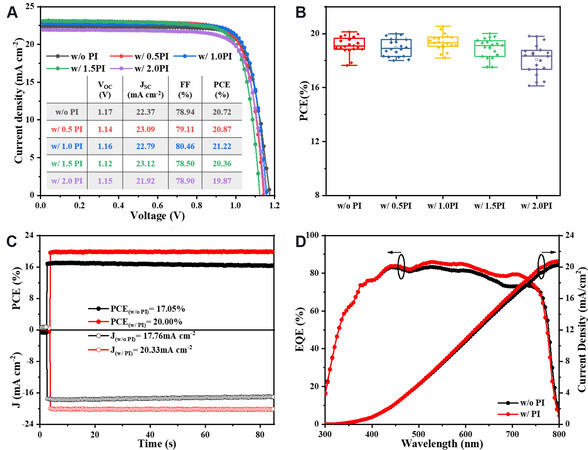
Figure 2. (A) J-V curves and (B) the PCE statistical distributions of 20 devices with different concentrations of PI. (C) The steady-state photocurrent measurements at maximum power point. (D) EQE spectra for the devices with (w/) and without (w/o) PI. PCE: High power conversion efficiency; EQE: external quantum efficiency; PI: polyimide.
The statistical PCE distribution of 20 independent devices fabricated from PI solution shown in Figure 2B suggests the same trend as the J-V curves. Besides, the devices based on 1.0 wt% PI exhibit the smallest variation and the best reproducibility. Therefore, it was chosen to compare with the PI-free device to the device with PI in the subsequent characterization, denoted as w/o PI and w/ PI, respectively.
To further confirm the values obtained from J-V curves, steady-state PCE was recorded over 85 s under the maximum power point voltage (0.98 and 0.96 V for devices w/ PI and w/o PI), as shown in Figure 2C, giving a PCE of 20.00% with the stabilized JSC of 20.33 mA/cm2 and a PCE of 17.05% with the stabilized JSC of 17.76 mA/cm2. Furthermore, the integrated JSC from EQE curves of devices w/ PI and w/o PI in Figure 2D are 20.76 and 20.29 mA/cm2, respectively, which agrees well with the J-V curves.
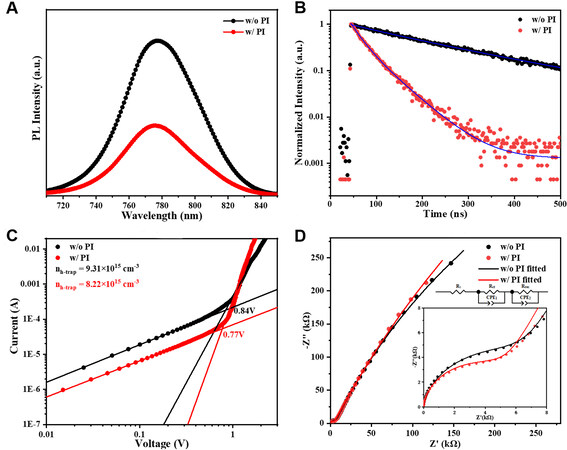
The dynamics of photo-excited carriers was investigated to understand the origin of the PCE enhancement after inserting this high-dielectric PI by PL and TRPL measurement, as shown in Figure 3A. A strong luminescence peak at 780 nm was observed, which was significantly decreased upon the insertion of PI interface, demonstrating the improved charge transfer efficiency. In addition, the PL lifetime was obtained by fitting the TRPL [Figure 3B] spectra with a two-exponential decay model: I(t) = A1exp(-t/τ1) + A2exp(-t/τ2) + I0. The fast decay lifetimes (τ1) represent the nonradiative recombination induced by the trapping process when the charge goes through the perovskite surface, and the slow decay lifetimes (τ2) are related to the radiative recombination process occurring in the bulk perovskite[29-31]. The fitted parameters are summarized in Supplementary Table 1. In this case, the average charge lifetime (τavg) was estimated to be 220.9 and
Figure 3. (A) PL spectra and (B) TRPL decay curves for the devices with (w/) and without (w/o) PI. (C) The dark current-voltage curves for hole-only devices with (w/) and without (w/o) PI. (D) Nyquist plots of the devices with (w/) and without (w/o) PI, measured in the dark. TRPL: Time-resolved photoluminescence; PI: polyimide; PL: photoluminescence.
Subsequently, to assess the hole-trap density at the perovskite/spiro-OMeTAD interfaces modified by PI layer, the SCLC method was conducted based on the hole-only devices, FTO/PEDOT:PSS/perovskite/with or without PI layer/spiro-OMeTAD/Ag. As shown in Figure 3C, the ohmic conducting region at lower voltage and trap-filled limited (TFL) region at higher voltage intersect at one point, where its corresponding voltage is the trap-filled limit voltage (VTFL)[33], which is reported positively correlated with the trap density
Moreover, from the Nyquist plots of PSCs in Figure 3D, obtained by EIS measurement in the dark, the semi-circle of the device w/ PI exhibits a smaller radius in the high frequency region, which demonstrates a lower charge transfer resistance (Rct). Simultaneously, the radius of its semi-circle in the low frequency region associated with the recombination resistance (Rrec)[35] is bigger than that of device w/o PI, consistent with the conclusion that the trap was mitigated by the PI interfaces.
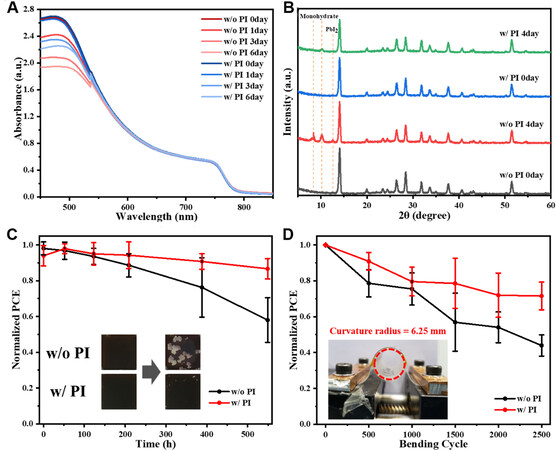
Finally, the influence of PI interfaces on the moisture and mechanical stability is shown in Figure 4. Firstly, MAPbI3 was deposited on the FTO substrate, followed by the deposition of the PI polymer. After a certain exposure time under high RH of 75% at 25 °C, the degree of degradation could be observed from the UV-vis absorption curve, as shown in Figure 4A. Both films showed a similar high absorbance on the first day, indicating that the introduction of PI layer has little effect on light absorption. After six days of exposure, the absorbance of the perovskite film w/o PI dropped sharply, while the PI-modified film gave little change. Simultaneously, XRD measurement was conducted to trace the phase transition of perovskite films. Figure 4B shows that the fresh w/o PI and w/ PI films presented main diffraction peaks of perovskite at 14.12°. However, in the spectra of aged perovskite film w/o PI (75% RH for four days), obvious peaks at 12.57°, 10.17°, and 8.40°, ascribed to PbI2 and monohydrate perovskite, were detected[11,36], forming a sharp contrast with the film w/ PI. Thereby, it is considered that the insertion of the PI layer indeed protects the perovskite film from fast moisture intrusion in high-humidity environments.
Figure 4. (A) Variations of the UV-vis absorption and (B) XRD measurement of perovskite without and with PI under an RH of 75% at 25 °C in dark conditions with respect to the exposure duration. (C) Long-term stability of the six corresponding PSCs under an RH of 50% at 25 °C in dark conditions. The inserts are optical photographs. (D) Normalized average PCE of 10 PSCs as a function of stretching cycles under a curvature radius of 6.25 mm. The inset shows the photograph of the flexible device under curvature. XRD: X-ray diffraction RH: relative humidity; PSCs: perovskite solar cells; PI: polyimide.
The long-term stability of PSCs in ambient condition was evaluated by monitoring the PCE of the devices without any encapsulation under 50% RH at 25 °C. As shown in Figure 4C, with PI treatment, the devices maintained 87% of their original PCE after aging over 550 h, whereas the PCE of the pristine perovskite films dropped to 58% of initial PCE. The insert figures show the optical photos of perovskite films stored in the same condition, where transparent hydrate can be found in the PI-free film after aging.
Aiming to validate the mechanical stability of the devices, the flexible PSCs were fabricated based on polyethylene naphthalate (PEN)/ITO substrate with or without PI, which showed a PCE of 17.52% and 16.90%, respectively [Supplementary Figure 6]. Figure 4D shows the normalized average PCE versus bending cycle under a curvature radius of 6.25 mm. The PI-modified devices exhibit remarkable stretching tolerance, maintaining 72% of the original PCE after 2500 cycles, while the PSCs without PI only gave 44% of the pristine PCE, possibly due to the formation of crack on the perovskite film[37]. Supplementary Table 2 shows a comparison of the moisture stability and mechanical stability of PSCs assisted by polymers, where our work shows outstanding moisture and mechanical stability. Moreover, our PI presents a simultaneous enhancement of both aspects. Thus, it is concluded that the high tolerance of mechanical stress of PI on top of perovskite could take part in the applied mechanical extrusion, giving rise to the mitigated influence of external mechanical stress.
CONCLUSIONS
In summary, we introduced a soluble PI as an interfacial layer to enhance the moisture and mechanical resistance. Further investigations manifested that the PI layer not only extracted holes but also reduced trap-assisted recombination at the interface, achieving a PCE of 21.22% in MAPbI3-based PSCs. Most importantly, at 50% RH, the devices with PI layer maintained 87% of the initial PCE after aging over 550 h, far superior to the pristine solar cells. Moreover, PI insertion enhanced the device tolerance against the external mechanical stress, providing an effective strategy to improve the stability of flexible PSCs.
DECLARATIONS
AcknowledgmentsThe authors gratefully acknowledge the funding support by NSFC Funds (U1801256, 51803064), Science and Technology Programs of Guangzhou (2019050001, 202002030130), International Cooperation Projects of Guangdong Province (2020A0505100054), Guangdong Provincial Key Laboratory of Optical Information Materials and Technology (2017B030301007), and Collaborative Research Fund (CRF C7018-20GF) of Hong Kong Research Grants Council. We also thank the support from the Guangdong Provincial Engineering Technology Research Center for Transparent Conductive Materials, and the Open Fund of the Key Laboratory of Functional Molecular Engineering of Guangdong Province (2018kfxx, South China University of Technology).
Authors’ contributionsContributions to conception and design of the study: Li Z, Jiang Y, Gao J
Data analysis and interpretation and data acquisition: Li Z, Kong X, Jiang Y, Gao J, Chen Y, Tan C
Administrative, technical, and material support: Gao J, Gao X, Lu X, Liu JM, Zhou G
Manuscript writing: Li Z, Jiang Y, Gao J
Availability of data and materialsNot applicable.
Financial support and sponsorshipNational Natural Science Foundation of China.
Conflicts of interestAll authors declared that there are no conflicts of interest.
Ethical approval and consent to participateNot applicable.
Consent for publicationNot applicable.
Copyright© The Author(s) 2021.
REFERENCES
1. Zhang F, Zhu K. Additive engineering for efficient and stable perovskite solar cells. Adv Energy Mater 2020;10:1902579.
2. Chen J, Park N. Materials and methods for interface engineering toward stable and efficient perovskite solar cells. ACS Energy Lett 2020;5:2742-86.
3. Ran C, Xu J, Gao W, Huang C, Dou S. Defects in metal triiodide perovskite materials towards high-performance solar cells: origin, impact, characterization, and engineering. Chem Soc Rev 2018;47:4581-610.
4. Zhao J, Xu Z, Zhou Z, et al. A safe flexible self-powered wristband system by integrating defective MnO2-x nanosheet-based Zinc-Ion batteries with perovskite solar cells. ACS Nano 2021;15:10597-608.
5. National Renewable Energy Laboratory USA, Best research-cell efficiencies. Available from: https://www.nrel.gov/pv/cell-efficiency.html. [Last accessed on 9 Aug 2021].
6. Jiang Q, Zhao Y, Zhang X, et al. Surface passivation of perovskite film for efficient solar cells. Nat Photonics 2019;13:460-6.
7. Yoo JJ, Seo G, Chua MR, et al. Efficient perovskite solar cells via improved carrier management. Nature 2021;590:587-93.
8. Wang R, Mujahid M, Duan Y, Wang Z, Xue J, Yang Y. A review of perovskites solar cell stability. Adv Funct Mater 2019;29:1808843.
9. Niu T, Lu J, Munir R, et al. Stable high-performance perovskite solar cells via grain boundary passivation. Adv Mater 2018;30:e1706576.
10. Choi K, Lee J, Kim HI, et al. Thermally stable, planar hybrid perovskite solar cells with high efficiency. Energy Environ Sci 2018;11:3238-47.
11. Leguy AMA, Hu Y, Campoy-quiles M, et al. Reversible hydration of CH3NH3PbI3 in films, single crystals, and solar cells. Chem Mater 2015;27:3397-407.
12. Xie Y, Cheng J, Liu H, et al. Co-Ni alloy@carbon aerogels for improving the efficiency and air stability of perovskite solar cells and its hysteresis mechanism. Carbon 2019;154:322-9.
13. Zheng H, Liu G, Zhu L, et al. The effect of hydrophobicity of ammonium salts on stability of quasi-2D perovskite materials in moist condition. Adv Energy Mater 2018;8:1800051.
14. Li F, Zhang J, Jo S, et al. Vertical orientated Dion-Jacobson quasi-2D perovskite film with improved photovoltaic performance and stability. Small Methods 2020;4:1900831.
15. Wang Z, Lin Q, Chmiel FP, Sakai N, Herz LM, Snaith HJ. Efficient ambient-air-stable solar cells with 2D-3D heterostructured butylammonium-caesium-formamidinium lead halide perovskites. Nat Energy 2017;2:17135.
16. Zhang T, Dar MI, Li G, et al. Bication lead iodide 2D perovskite component to stabilize inorganic α-CsPbI3 perovskite phase for high-efficiency solar cells. Sci Adv 2017;3:e1700841.
17. Kim M, Motti SG, Sorrentino R, Petrozza A. Enhanced solar cell stability by hygroscopic polymer passivation of metal halide perovskite thin film. Energy Environ Sci 2018;11:2609-19.
18. Zhang H, Shi J, Zhu L, et al. Polystyrene stabilized perovskite component, grain and microstructure for improved efficiency and stability of planar solar cells. Nano Energy 2018;43:383-92.
19. Yu Y, Tseng C, Chien W, Hsu H, Chen C. Photovoltaic performance enhancement of perovskite solar cells using polyimide and polyamic acid as additives. J Phys Chem C 2019;123:23826-33.
20. Jeong G, Koo D, Seo J, et al. Suppressed interdiffusion and degradation in flexible and transparent metal electrode-based perovskite solar cells with a graphene interlayer. Nano Lett 2020;20:3718-27.
21. Yoon J, Kim U, Yoo Y, et al. Foldable perovskite solar cells using carbon nanotube-embedded ultrathin polyimide conductor. Adv Sci (Weinh) 2021;8:2004092.
22. Chen C, Jiang Y, Guo J, et al. Solvent-assisted low-temperature crystallization of SnO2 electron-transfer layer for high-efficiency planar perovskite solar cells. Adv Funct Mater 2019;29:1900557.
23. Zhang S, Liu Z, Zhang W, et al. Barrier designs in perovskite solar cells for long-term stability. Adv Energy Mater 2020;10:2001610.
24. Wang Q, Dong Q, Li T, Gruverman A, Huang J. Thin insulating tunneling contacts for efficient and water-resistant perovskite solar cells. Adv Mater 2016;28:6734-9.
25. Wen X, Wu J, Ye M, Gao D, Lin C. Interface engineering via an insulating polymer for highly efficient and environmentally stable perovskite solar cells. Chem Commun (Camb) 2016;52:11355-8.
26. Yang Z, Ma P, Li F, Guo H, Kang C, Gao L. Ultrahigh thermal-stability polyimides with low CTE and required flexibility by formation of hydrogen bonds between poly(amic acid)s. European Polymer Journal 2021;148:110369.
27. Zhang J, Jiang P, Wang Y, Liu X, Ma J, Tu G. In situ synthesis of ultrastable CsPbBr3 perovskite nanocrystals coated with polyimide in a CSTR system. ACS Appl Mater Interfaces 2020;12:3080-5.
28. Chen N, Yi X, Zhuang J, et al. An efficient trap passivator for perovskite solar cells: poly(propylene glycol) bis(2-aminopropyl ether). Nanomicro Lett 2020;12:177.
29. Huang Z, Hu X, Liu C, et al. Water-resistant and flexible perovskite solar cells via a glued interfacial layer. Adv Funct Mater 2019;29:1902629.
30. Cai Y, Cui J, Chen M, et al. Multifunctional enhancement for highly stable and efficient perovskite solar cells. Adv Funct Mater 2021;31:2005776.
31. Liu K, Liang Q, Qin M, et al. Zwitterionic-surfactant-assisted room-temperature coating of efficient perovskite solar cells. Joule 2020;4:2404-25.
32. Chen C, Li F, Zhu L, et al. Efficient and stable perovskite solar cells thanks to dual functions of oleyl amine-coated PbSO4(PbO)4 quantum dots: Defect passivation and moisture/oxygen blocking. Nano Energy 2020;68:104313.
33. Duijnstee EA, Ball JM, Le Corre VM, Koster LJA, Snaith HJ, Lim J. Toward understanding space-charge limited current measurements on metal halide perovskites. ACS Energy Lett 2020;5:376-84.
34. Liu Z, Cao F, Wang M, Wang M, Li L. Observing defect passivation of the grain boundary with 2-aminoterephthalic acid for efficient and stable perovskite solar cells. Angew Chem Int Ed Engl 2020;59:4161-7.
35. Zhao Y, Zhu P, Huang S, et al. Molecular interaction regulates the performance and longevity of defect passivation for metal halide perovskite solar cells. J Am Chem Soc 2020;142:20071-9.
36. Yun S, Ma S, Kwon H, et al. Amino acid salt-driven planar hybrid perovskite solar cells with enhanced humidity stability. Nano Energy 2019;59:481-91.
Cite This Article
Export citation file: BibTeX | RIS
OAE Style
Li Z, Kong X, Jiang Y, Lu X, Gao X, Tan C, Chen Y, Zhou G, Liu JM, Gao J. Simultaneously enhancing moisture and mechanical stability of flexible perovskite solar cells via a polyimide interfacial layer. Soft Sci 2021;1:4. http://dx.doi.org/10.20517/ss.2021.06
AMA Style
Li Z, Kong X, Jiang Y, Lu X, Gao X, Tan C, Chen Y, Zhou G, Liu JM, Gao J. Simultaneously enhancing moisture and mechanical stability of flexible perovskite solar cells via a polyimide interfacial layer. Soft Science. 2021; 1(1): 4. http://dx.doi.org/10.20517/ss.2021.06
Chicago/Turabian Style
Li, Zhuoxi, Xiangyu Kong, Yue Jiang, Xubing Lu, Xinseng Gao, Chaoliang Tan, Yiwang Chen, Guofu Zhou, Jun-Ming Liu, Jinwei Gao. 2021. "Simultaneously enhancing moisture and mechanical stability of flexible perovskite solar cells via a polyimide interfacial layer" Soft Science. 1, no.1: 4. http://dx.doi.org/10.20517/ss.2021.06
ACS Style
Li, Z.; Kong X.; Jiang Y.; Lu X.; Gao X.; Tan C.; Chen Y.; Zhou G.; Liu J.M.; Gao J. Simultaneously enhancing moisture and mechanical stability of flexible perovskite solar cells via a polyimide interfacial layer. Soft. Sci. 2021, 1, 4. http://dx.doi.org/10.20517/ss.2021.06
About This Article
Copyright
Data & Comments
Data
 Cite This Article 12 clicks
Cite This Article 12 clicks














Comments
Comments must be written in English. Spam, offensive content, impersonation, and private information will not be permitted. If any comment is reported and identified as inappropriate content by OAE staff, the comment will be removed without notice. If you have any queries or need any help, please contact us at support@oaepublish.com.